Check the below NCERT MCQ Questions for Class 10 Sanskrit Chapter 6 सुभाषितानि with Answers Pdf free download. MCQ Questions for Class 10 Sanskrit with Answers were prepared based on the latest exam pattern. We have provided सुभाषितानि Class 10 Sanskrit MCQs Questions with Answers to help students understand the concept very well.
Students can also read NCERT Solutions for Class 10 Sanskrit Chapter 6 Questions and Answers at LearnInsta. Here all questions are solved with a detailed explanation, It will help to score more marks in your examinations.
उचित पदं चित्वा प्रश्ननिर्माणं कुरुत
Question 1.
अस्तमये सविता रक्तः भवति।
(i) के
(ii) कदा
(iii) कस्मै
(iv) कुत्र
Answer
Answer: (ii) कदा
Question 2.
वायसः वसन्तस्य गुणं न जानाति।
(i) कः
(ii) कस्य
(iii) केन
(iv) कदा
Answer
Answer: (ii) कस्य
Question 3.
यः निमित्तम् उद्दिश्य प्रकुप्यति।
(i) किम्
(ii) कः
(iii) कथम्
(iv) केन
Answer
Answer: (i) किम्
Question 4.
क्रोधः देहविनाशनाय प्रथमः शत्रुः।
(i) कः
(ii) कस्मै
(iii) कस्य
(iv) काय
Answer
Answer: (ii) कस्मै
Question 5.
गुणी गुणं वेत्ति।
(i) कः
(ii) की
(iii) का
(iv) के
Answer
Answer: (i) कः
Question 6.
महत्ताम् एकरूपता संपत्तौ विपत्तौ च भवति।
(i) कदा
(ii) कुत्र
(iii) कौ
(iv) के
Answer
Answer: (i) कदा
Question 7.
फलच्छायासमन्वितः महा वृक्षः सेवितव्यः।
(i) का
(ii) कीदृशः
(iii) कया
(iv) कः
Answer
Answer: (ii) कीदृशः
Question 8.
मूर्खा मूखैः सह अनुव्रजन्ति।
(i) केन
(ii) कैः
(iii) के
(iv) कदा
Answer
Answer: (ii) कैः
Question 9.
पशुना अपि उदीरितः अर्थः गृह्यते।
(i) कैः
(ii) केन
(iii) काभिः
(iv) कया
Answer
Answer: (ii) केन
Question 10.
सिंहस्य बलं गजः वेत्ति मूषकः न।
(i) कस्य
(ii) कः
(iii) किम्
(iv) कस्याः
Answer
Answer: (i) कस्य
Question 11.
संसारे किञ्चित् निरर्थकम् न अस्ति।
(i) कैः
(ii) कुत्र
(iii) कस्मै
(iv) किम्
Answer
Answer: (ii) कुत्र
Question 12.
क्रोधः नरस्य प्रथमः शत्रुः।
(i) कीदृशः
(ii) कीदृशम्
(iii) कः
(iv) के
Answer
Answer: (i) कीदृशः
Question 13.
अक्षरं अमन्त्रम् नस्ति।
(i) कीदृशम्:
(ii) कीदृशः
(iii) किम्
(iv) कम्
Answer
Answer: (i) कीदृशम्:
Question 14.
महावृक्षस्य छाया केन निवार्यते।
(i) का
(ii) कः
(iii) किम्
(iv) कीदृशः
Answer
Answer: (i) का
अधोलिखितं पद्यांशं पठित्वा निर्देशानुसार प्रश्नान् उत्तरत
(क) आलस्यं हि मनुष्याणां शरीरस्थो महान् रिपुः।
नास्त्युद्यमसमो बन्धुः कृत्वा यं नावसीदति॥
Question 1.
अस्माकं महान् रिपुः कः अस्ति?
Answer
Answer: आलस्यम्
Question 2.
आलस्यं कुत्र स्थितः अस्ति?
Answer
Answer: शरीरे
Question 3.
आलस्यं कृत्वा कः अवसीदति?
Answer
Answer: मनुष्यः
Question 4.
किं कृत्वा मनुष्यः न अवसीदति?
Answer
Answer: उद्यमं कृत्वा मनुष्यः न अवसीदति।
Question 5.
कः मनुष्याणां बन्धुसमः अस्ति?
Answer
Answer: उद्यमः मनुष्याणां बन्धुसमः अस्ति।
Question 6.
‘महान् रिपुः’ अनयोः पदयोः विशेषणपदं किम्?
Answer
Answer: महान्
Question 7.
‘नावसीदति’ इति क्रियापदस्य कर्तृपदम् किम्?
Answer
Answer: मनुष्यः
Question 8.
‘उद्यमम्’ इति पदस्य विपर्ययदं पद्यांशे किमस्ति?
Answer
Answer: आलस्यं
Question 9.
श्लोके ‘शत्रुः’ इत्यस्य पदस्य कः पर्यायः आगतः?
Answer
Answer: रिपुः
(ख) गुणी गुणं वेत्ति न वेत्ति निर्गणो,
बली बलं वेत्ति न वेत्ति निर्बलः।
पिको वसन्तस्य गुणं न वायसः,
करी च सिंहस्य बलं न मूषकः॥
Question 1.
कीदृशः जनः गुणं जानाति?
Answer
Answer: गुणी
Question 2.
कीदृशः जनः गुणं न जानाति?
Answer
Answer: निर्गुणः
Question 3.
करी कस्य बलं जानाति?
Answer
Answer: सिंहस्य
Question 4.
पिकः किम् जानाति वायसः च किम् न?
Answer
Answer: पिक: वसन्तस्य गुणं जानाति वायसः च वसन्तस्य गुणं न जानाति।
Question 5.
मूषकः कस्य बलं न जानाति?
Answer
Answer: मूषकः सिंहस्य बलं न जानाति।
Question 6.
श्लोके ‘जानाति’ इति पदस्य पर्यायपदं किम् आगतम्?
Answer
Answer: वेत्ति
Question 7.
‘वेत्ति’ इति क्रियापदस्य कर्तृपदं श्लोके किम्?
Answer
Answer: गुणी
Question 8.
‘गजः’ इत्यर्थे किं पदं पद्यांशे प्रयुक्तम्?
Answer
Answer: करी
Question 9.
श्लोके ‘दुर्गुणम्’ इत्यस्य पदस्य कः विपर्ययः लिखितः?
Answer
Answer: गुणम्
(ग) निमित्तमुद्दिश्य हि यः प्रकुप्यति,
ध्रुवं स तस्यापगमे प्रसीदति।
अकारणद्वेषि मनस्तु यस्य वै,
कथं जनस्तं परितोषयिष्यति॥
Question 1.
नरः किम् उद्दिश्य प्रकुप्यति?
Answer
Answer: निमित्तम्
Question 2.
नरः कदा प्रसीदति?
Answer
Answer: तस्यापगमे (निमित्तापगमे)
Question 3.
कदाचित् अकारणद्वेषि किं भवति?
Answer
Answer: मनः
Question 4.
यस्य मनुष्यस्य मनः अकारणद्वेषि अस्ति किं सः कदापि परितोषयिष्यति?
Answer
Answer: यस्य मनुष्यस्य मनः अकारणदेषि अस्ति तं जनः कदापि न परितोषयिष्यति।
Question 5.
कः निमित्तम् उद्दिश्य प्रकुप्यति?
Answer
Answer: जनः निमित्तमुदिश्य प्रकुप्यति?
Question 6.
‘यः’ पदं पद्यांशे कस्मै प्रयुक्तम्?
Answer
Answer: नराय
Question 7.
श्लोके ‘प्रकुप्यति’ इति क्रिया पदस्य कर्तृपदं किम्?
Answer
Answer: यः
Question 8.
‘सः’ इति कर्तृपदस्य क्रियापदं किम्?
Answer
Answer: प्रसीदति
Question 9.
श्लोके ‘कारणम्’ इत्यस्य पदस्य कः पर्यायः आगतः?
Answer
Answer: निमित्तम्
(घ) उदीरितोऽर्थः पशुनापि गृह्यते,
ह्याश्च नागाश्च वहन्ति बोधिताः।
अनुक्तमप्यूहति पण्डितो जनः,
परेङ्गितज्ञानफला हि बुद्धयः ॥ 4 ॥
Question 1.
उदीरितोऽर्थः केनापि गृह्यते?
Answer
Answer: पशुना
Question 2.
केऽपि वहन्ति बोधिता:?
Answer
Answer: नागा:/हयाः
Question 3.
पण्डितः जनः किमपि ऊहति?
Answer
Answer: अनुक्तम्
Question 4.
बुद्धयः कीदृश्यः भवन्ति?
Answer
Answer: बुद्धयः परेङ्गितज्ञानफलाः हि भवन्ति।
Question 5.
पशुनापि कः गृह्यते?
Answer
Answer: पशुनापि उदीरितोऽर्थः गृह्यते।
Question 6.
अत्र श्लोके ‘गृह्यते’ क्रियापदस्य कर्तृपदं किम्?
Answer
Answer: अर्थः
Question 7.
‘विद्वान् जनः’ इत्यर्थे किं पदं प्रयुक्तम्?
Answer
Answer: पण्डितो जनः
Question 8.
‘अश्वाः ‘ इति पदस्य पर्यायपदं पद्यांशे किम्?
Answer
Answer: हयाः
Question 9.
श्लोके ‘मुच्यते’ इति क्रियापदस्य कः विपर्ययः?
Answer
Answer: गृह्यते
(ङ) क्रोधो हि शत्रुः प्रथमो नराणां,
देहस्थितो देहविनाशनाय।
यथास्थितः काष्ठगतो हि वह्निः,
स एव वह्निर्दहते शरीरम्॥
Question 1.
नराणां प्रथमः शत्रुः कः?
Answer
Answer: क्रोधः
Question 2.
क्रोधः केषाम् प्रथमः शुत्रः?
Answer
Answer: नराणाम्
Question 3.
क्रोधः किमर्थं भवति?
Answer
Answer: देहविनाशनाय
Question 4.
कीदृशो अग्निः शरीराणि दहति?
Answer
Answer: क्रोधरूपाग्निः शरीराणि दहति।
Question 5.
शरीरं कः दहते?
Answer
Answer: शरीरं क्रोधः दहते।
Question 6.
‘प्रथमः शत्रुः’ अनयो:पदयोः विशेषणपदं किम्?
Answer
Answer: प्रथमः
Question 7.
‘दहते’ इति क्रियापदस्य कर्ता क:?
Answer
Answer: वह्निः
Question 8.
‘रक्षणाय’ इति पदस्य विपर्ययपदं किम्?
Answer
Answer: विनाशनाय
Question 9.
श्लोके ‘अग्निः’ इति पदस्य कः पर्याय आगन:?
Answer
Answer: वह्निः
(च) मृगाः मृगैः सङ्गमनुव्रजन्ति,
गावश्च गोभिः तुरगास्तुरङ्गैः।
मूर्खाश्च मूखैः सुधियः सुधीभिः,
समान-शील-व्यसनेषु सख्यम्॥
Question 1.
मृगाः कैः सङ्गमनुव्रजन्ति?
Answer
Answer: मृगैः
Question 2.
मूखैः सह के अनुव्रजन्ति?
Answer
Answer: मूर्खाः
Question 3.
तुरगाः कैः सह अनुव्रजन्ति?
Answer
Answer: तुरङ्गैः
Question 4.
किम् समान-शील-व्यसनेषु भवति?
Answer
Answer: सख्यम् समान-शील-व्यसनेषु भवति?
Question 5.
सुधीभिः सह के अनुगच्छन्ति?
Answer
Answer: सुधीभिः सह सुधियः अनुगच्छन्ति।
Question 6.
‘अनुव्रजन्ति’ इति क्रियापदस्य कर्तृपदं किम्?
Answer
Answer: मृगाः
Question 7.
‘तुरगा’ इति कर्तृपदस्य क्रियापदं किम्?
Answer
Answer: अनुव्रजन्ति
Question 8.
‘सुधीभिः’ इति पदस्य विपर्ययपदं किं प्रयुक्तम्?
Answer
Answer: मूर्खः
Question 9.
‘अश्वाः’ इति पदस्य कः पर्यायः श्लोके आगतः?
Answer
Answer: तुरगाः
(छ) सेवितव्यो महावृक्षः फलच्छायासमन्वितः।
यदि दैवात् फलं नास्ति छाया केन निवार्यते॥
Question 1.
कीदृशः वृक्षः सेवितव्यः?
Answer
Answer: महावृक्षः
Question 2.
महावृक्षः कीदृशः भवति?
Answer
Answer: फलच्छायासमन्वितः
Question 3.
वृक्षेषु का भवति?
Answer
Answer: छाया
Question 4.
महावृक्षेषु दैवात् फलं नास्ति तथापि किं न निवार्यते?
Answer
Answer: महावृक्षेषु दैवात् फलं नास्ति तथापि छाया केनापि न निवार्यते।
Question 5.
फलच्छायासमन्वितः कः सेवितव्यः?
Answer
Answer: फलच्छायासमन्वितः महावृक्षः सेवितव्यः।
Question 6.
‘महान् वृक्षः’ अत्र विशेष्यपदं किम्?
Answer
Answer: वृक्षः
Question 7.
‘भाग्यवशात्’ इत्यर्थे किं पदं प्रयुक्तम्?
Answer
Answer: दैवात्
Question 8.
‘लघुवृक्षः’ इति पदस्य विपर्ययपदं किं प्रयुक्तम्?
Answer
Answer: महावृक्षः
Question 9.
‘छाया केन निवार्यते’ अत्र कर्तृपदं किम्?
Answer
Answer: केन
(ज) अमन्त्रमक्षरं नास्ति, नास्ति मूलमनौषधम्।
अयोग्यः पुरुषः नास्ति योजकस्तत्र दुर्लभः॥
Question 1.
अक्षरं कीदृशं न भवति?
Answer
Answer: मन्त्ररहितम्
Question 2.
मूलम् कीदृशम् न भवति?
Answer
Answer: अनौषधम्
Question 3.
संसारे कः दुर्लभो भवति?
Answer
Answer: योजकः
Question 4.
कीदृशः पुरुषः वास्तविकः पुरुषः न भवति?
Answer
Answer: अयोग्यः पुरुषः वास्तविकः पुरुषः न भवति।
Question 5.
अमन्त्रं किं न भवति?
Answer
Answer: अमन्त्रम् अक्षरं न भवति।
Question 6.
‘अमन्त्रं अक्षरं’ अनयोः पदयोः विशेषणपदं किम्?
Answer
Answer: अमन्त्रं
Question 7.
‘अयोग्यः’ इति पदस्य विशेष्यपदं किम्?
Answer
Answer: पुरुषः
Question 8.
‘सुलभः’ इति पदस्य विपर्ययपदं पद्यांशे किमस्ति?
Answer
Answer: दुर्लभः
Question 9.
‘नास्ति मूलम् अनौषधम्’। अत्र क्रियापदं किम्?
Answer
Answer: नास्ति
(झ) संपत्तौ च विपत्तौ च महतामेकरूपता।
उदये सविता रक्तो रक्तोश्चास्तमये तथा॥
Question 1.
उदये सति सूर्यः कीदृशः भवति?
Answer
Answer: रक्तः
Question 2.
अस्तमये च सवितुः वर्णः कीदृशः भवति?
Answer
Answer: रक्तः
Question 3.
केषाम् एकरूपता भवति?
Answer
Answer: महताम्
Question 4.
महताम् एकरूपता कदा भवति?
Answer
Answer: महताम् संपत्तौ विपत्तौ च एकरूपता भवति।
Question 5.
उदये अस्तमये च सविता कीदृशो भवति?
Answer
Answer: उदये अस्तमये च सविता रक्तः भवति।
Question 6.
‘संपत्तौ’ पदस्य विपर्ययपदं पद्यांशे किमस्ति?
Answer
Answer: विपत्तौ
Question 7.
‘सूर्य:’ पदस्य पर्यायपदं किम्?
Answer
Answer: सविता
Question 8.
‘भवति’ इति क्रियापदस्य कर्तृपदं किम्?
Answer
Answer: सविता
Question 9.
श्लोके ‘सविता रक्तः’ अनयोः पदयोः किं विशेषणम्?
Answer
Answer: रक्तः
(ञ) विचित्रे खलु संसारे नास्ति किञ्चिन्निरर्थकम्।
अश्वश्चेद् धावते वीरः भारस्य वहने खरः॥
Question 1.
संसारः कीदृशः अस्ति?
Answer
Answer: विचित्रः
Question 2.
म्निरर्थकम् कुत्र न अस्ति?
Answer
Answer: संसारे
Question 3.
भारवहने कः वीरः भवति?
Answer
Answer: खरः
Question 4.
अश्वः खरः च कीदृशौ वीरौ स्तः?
Answer
Answer: अश्वः चेत् धावने वीरः तर्हि भारस्य वहने खरः वीरः अस्ति।
Question 5.
विचित्रः खलु को वर्तते?
Answer
Answer: विचित्रः खलु संसारः वर्तते।
Question 6.
‘संसारे’ इति पदस्य विशेषणपदं किम्?
Answer
Answer: विचित्रे
Question 7.
‘सार्थकम्’ इति पदस्य विपर्ययपदं किमस्ति?
Answer
Answer: निरर्थकम्
Question 8.
‘नास्ति’ इति क्रियापदस्य कर्तृपदं किम्?
Answer
Answer: किञ्चित्
Question 9.
श्लोके ‘गर्दभः’ इत्यस्य पदस्य कः पर्यायः आगतः?
Answer
Answer: खरः
प्रश्ननिर्माणम् कुरुत
(क) आलस्यं मनुष्याणाम् शत्रुः।
(ख) आलस्यं मनुष्याणां शरीरस्थो महान् रिपुः।
(ग) उद्यतेनसमः बन्धु नास्ति।
(घ) नरः उद्यतं कृत्वा न अवसीदति।
(ङ) आलस्यं मनुष्याणां शरीरस्थो महान् रिपुः।
(च) बलवान् बलम् वेत्ति?
(छ) वसन्तस्य गुण: पिक: जानाति।
(ज) निर्गुणः गुणं न वेत्ति।
(झ) मृगाः मृगैः सङ्गमनुव्रजन्ति।
(ञ) गावः गोभिः सङ्गमनुव्रजन्ति।
(ट) नराणां प्रथमः शुत्रः क्रोधः अस्ति।
(ठ) फलच्छायासमन्वितः महावृक्षः सेवितव्यः।
(ड) संपत्तौ विपत्तौ च महताम् एकरूपता भवति।
(ढ) सविता उदयेसति रक्तः भवति।
(ण) एषः संसारः विचित्रः अस्ति।
(त) अत्र किञ्चिन्निरर्थकम् न अस्ति।
Answer
Answer:
(क) केषाम्
(ख) किम्
(ग) केन
(घ) किम्
(ङ) कीदृशः
(च) कः
(छ) कः
(ज) कः
(झ) के
(ञ) काभिः
(ट) केषाम्
(ठ) कीदृशः
(ड) केषाम्
(ढ) कदा
(ण) कीदृशः
(त) कीदृशम्
अन्वयं लेखनम् श्लोकस्य अन्वयं मञ्जूषायाः सहायता पूरयन्तु
(क) आलस्यं हि मनुष्याणां शरीरस्थो महान् रिपुः।
नास्त्युद्यमसमो बन्धुः कृत्वा यं नावसीदति॥
अव्ययः-मनुष्याणां (i) ………………… महान् शत्रुः (ii) ……………. । उद्यमसमः (iii) ……………… न अस्ति यं (iv) ………………… (मनुष्यः) न अवसीदति।
मञ्जूषा- कृत्वा, शरीरस्थः, बन्धुः, आलस्यम्
Answer
Answer:
(i) शरीरस्थः
(ii) आलस्यम्
(iii) बन्धुः
(iv) कृत्वा
(ख) गुणी गुणं वेत्ति न वेत्ति निर्गुणो,
बली बलं वेत्ति न वेत्ति निर्बलः।
पिको वसन्तस्य गुणं न वायसः,
करी च सिंहस्य बलं न मूषकः॥
अव्ययः- गुणी गुणं (i) ………….. निर्गुणः (गुणं) न वेत्ति, (ii) ……………… बलं वेत्ति (iii) ………………. (बल) न वेत्ति, वसन्तस्य गुणं पिकः (वेत्ति), (iv) ……………..
न (वेत्ति), सिंहस्य बलं करी (वेत्ति) (v) ………………. न।
मञ्जूषा- वायसः, वेत्ति, मूषकः, बली, निर्बलः
Answer
Answer:
(i) वेत्ति
(ii) बली
(iii) निर्बलः
(iv) वायसः
(v) मूषक:
निमित्तमुद्दिश्य हि यः प्रकुप्यति,
ध्रुवं स तस्यापगमे प्रसीदति।
अकारणद्वेषि मनस्तु यस्य वै,
कथं जनस्तं परितोषयिष्यति॥
अव्ययः- यः निमित्तम् (i) ………….. प्रकुप्यति सः तस्य (ii) …………….. ध्रुवं प्रसीदति यस्य (iii) …………….. अकारणद्वेषि (अस्ति) जनः तं (iv) ……………. परितोषयिष्यति।
मञ्जूषा- अपगमे, कथम्, मनः, उद्दिश्य
Answer
Answer:
(i) उद्दिश्य
(ii) अपगमे
(iii) मनः
(iv) कथम्
(घ) उदीरितोऽर्थः पशुनापि गृह्यते,
हृयाश्च नागाश्च वहन्ति बोधिताः।
अनुक्तमप्यूहति पण्डितो जनः,
परेङ्गितज्ञानफला हि बुद्धयः॥
अन्वयः- पशुना अपि (i) …………….. अर्थः गृह्यते, (ii) …………….. नागाः च बोधिताः (भारं) (iii) …………….. पण्डितः जनः (iv) ……………. अपि ऊहति (v) ………….. परेङ्गितज्ञानफलाः भवन्ति।
मञ्जूषा- वहन्ति, बुद्धयः, उदीरितः, अनुक्तम्, हयाः
Answer
Answer:
(i) उदीरितः
(ii) हयाः
(iii) वहन्ति
(iv) अनुक्तम्
(v) बुद्धयः
(ङ) क्रोधो हि शत्रुः प्रथमो नराणां,
देहस्थितो देहविनाशनाय।
यथास्थितः काष्ठगतो हि वाह्निः,
स एव वह्निर्दहते शरीरम्॥
अन्वयः- नराणां देहविनाशनाय (i) ……………. शत्रः देहस्थितः (ii) ……………. । यथा काष्ठगतः स्थितः (iii) ………………. काष्ठम् एव (iv) …………… (तथैव शरीरस्थः क्रोधः) शरीरं दहते। मञ्जूषा- वह्निः, प्रथमः, दहते, क्रोधः
Answer
Answer:
(i) प्रथमः
(ii) क्रोधः
(iii) वह्निः
(iv) दहते
(च) मृगाः मृगैः सङ्गमनुव्रजन्ति,
गावश्च गोभिः तुरगास्तुरङ्गैः।
मूर्खाश्च मूर्खः सुधियः सुधीभिः,
समान-शील-व्यसनेषु सख्यम्॥
अन्वयः- मृगाः (i) ………………. सह, गावश्च गोभिः सह, (ii) …………….. तुरङ्गः सह, मूर्खाः मूर्खे (iii) …………… सुधियः सुधीभिः सह (iv) ……………… समान (v) …………….. व्यसनेषु सख्यम् (भवति)।
मञ्जूषा- सह, मृगैः, शील, तुरगाः, अनुव्रजन्ति
Answer
Answer:
(i) मृगैः
(ii) तुरगाः
(iii) सह
(iv) अनुव्रजन्ति
(v) शील
(छ) सेवितव्यो महावृक्षः फलच्छायासमन्वितः।
यदि दैवात् फलं नास्ति छाया केन निवार्यते॥
अन्वयः- फलच्छाया समन्वितः (i) …………….. सेवितव्यः (ii) …………….. यदि फलं (iii) …………….. (वृक्षस्य) (iv) …………… केन निवार्यते।
मञ्जूषा- छाया, महावृक्षः, नास्ति, दैवात्।
Answer
Answer:
(i) महावृक्षः
(ii) दैवात्
(iii) नास्ति
(iv) छाया
(ज) अमन्त्रमक्षरं नास्ति, नास्ति मूलमनौषमम्।
अयोग्यः पुरुषः नास्ति योजकस्तत्र दुर्लभः॥
अन्वयः- अमन्त्रम् (i) …………………. नास्ति (ii) ………….. मूलं नास्ति, (iii) ………………. पुरुष: नास्ति, तत्र (iv) …………….. दुर्लभः।
मञ्जूषा- अयोग्यः, अनौषधम्, योजकः, अक्षरं
Answer
Answer:
(i) अक्षरं
(ii) अनौषधम्
(iii) अयोग्यः
(iv) योजकः
(झ) संपत्तौ च विपत्तौ च महतामेकरूपता।
उदये सविता रक्तो रक्तोश्चास्तमये तथा॥
अन्वयः- महताम् (i) ………….. विपत्तौ च (ii) ………….. भवति (यथा) (iii) ……………… (iv) ………………. भवति, तथा (v) ………………. च रक्तः भवति।
मञ्जूषा- अस्तमये, संपत्तौ, सविता, एकरूपता, रक्तः
Answer
Answer:
(i) संपत्तौ
(ii) एकरूपता
(iii) सविता
(iv) रक्तः
(v) अस्तमये
(ञ) विचित्रे खलु संसारे नास्ति किञ्चिनिरर्थकम्।
अश्वश्चेद् धावने वीरः भारस्य वहने खरः॥
अन्वयः- विचित्रे (i) ……………. खलु किञ्चित् (ii) ……………. नास्ति। अश्वः चेत् (iii) ………………. वीरः (तर्हि) भारस्य वहने (iv) ………………. (वीरः) अस्ति।
मञ्जूषा- निरर्थकं, खरः, धावने, संसारे
Answer
Answer:
(i) संसारे
(ii) निरर्थकं
(iii) धावने
(iv) खरः
भावार्थलेखनम् श्लोकानाम् भावं उचितैः शब्दैः सम्पूरयत
(क) आलस्यं हि मनुष्याणां शरीरस्थो महान् रिपुः।
नास्त्युद्यमसमो बन्धुः कृत्वा यं नावसीदति॥
भावार्थ:- अस्य भावोऽस्ति यत् (i) …………….. शरीरे स्थितम् (ii) …………….. एव तेषां। महान् शत्रुः अस्ति। एवमेव पुरुषार्थस्य इव तेषां कोऽपि अन्यः (iii) ……………… नास्ति। तं (पुरुषार्थ) कृत्वा (iv) ………………. कदापि न दुखीयन्ति।
मञ्जूषा- मानवाः, मानवाना, शत्रुः, आलस्यम्
Answer
Answer:
(i) मानवानां
(ii) आलस्यम्
(iii) शत्रुः
(iv) मानवाः
(ख) गुणी गुणं वेत्ति न वेत्ति निर्गुणो,
बली बलं वेत्ति न वेत्ति निर्बलः।
पिको वसन्तस्य गुणं न वायसः,
करी च सिंहस्य बलं न मूषकः॥
भावार्थ:- संसारे गुणवान् जनः एव (i) …………….. महत्वं जानाति, गुणैः हीनः जनः न जानाति, (ii) ……………… एवं बलस्य महत्वं जानाति बलहीनः न जानाति। (iii) ……………. महत्वं कोकिलः एवं जानाति काकः न एवमेव सिंहस्य बलं तु गज एव जानाति (iv) ……………. तु कदापि न जानाति।
मञ्जूषा- बलवान्, मूषकः, गुणस्य, वसन्तस्य
Answer
Answer:
(i) गुणस्य
(ii) बलवान्
(iii) वसन्तस्य
(iv) मूषकः
(ग) निमित्तमुद्दिश्य हि यः प्रकुप्यति,
ध्रुवं स तस्यापगमे प्रसीदति।
अकारणद्वेषि मनस्तु यस्य वै,
कथं जनस्तं परितोषयिष्यति॥
भावार्थ:- यः जनः कदाचित् किञ्चित् (i) …………….. उद्दिश्य कस्मैचिदपि क्रुध्यति स एव तस्य कारणस्य (ii) ……………. जने प्रसीदति। एवमेव यस्य जनस्य (iii) …………. अकारणमेव केनचित् सह द्वेषं करोति तं जनं कथं (iv) ………………. कर्तुं समर्थाः भविष्यन्ति जनाः।
मञ्जूषा- तस्मिन्, मनः, कारणम्, सन्तुष्टम्
Answer
Answer:
(i) कारणम्
(ii) तस्मिन्
(iii) मनः
(iv) सन्तुष्टम्
(घ) उदीरितोऽर्थः पशुनापि गृह्यते,
हयाश्च नागाश्च वहन्ति बोधिताः।
अनुक्तमप्यूहति पण्डितो जनः,
परेगितज्ञानफला हि बुद्धयः॥
भावार्थ:- अस्य श्लोकस्य भावोऽस्ति यत् अश्वाः गजाश्च (i) ……………. अपि कृतं संकेतं ज्ञात्वा (ii) ……………… वहन्ति। तथैव विद्वान् जनः अकथितम् (iii) …………….. ज्ञात्वा कार्यं कुर्वन्ति यतः बुद्धयः परैः संकेतात् उत्पन्ना: (iv) …………….. वन्ति।
मञ्जूषा- फलदायिकाः, अन्यैः, भारं, संकेतम्
Answer
Answer:
(i) अन्यैः
(ii) भारं
(iii) संकेतम्
(iv) फलदायिकाः
(ङ) क्रोधो हि शत्रुः प्रथमो नराणां,
देहस्थितो देहविनाशनाय।
यथास्थितः काष्ठगतो हि वाह्निः,
स एव वह्निर्दहते शरीरम्॥
भावार्थ:- अर्थात् मनुष्याणां शरीरेषु स्थितः (i) ……………. एव तेषां देहानाम् विनाशस्य प्रथमः (ii) …………… अस्ति। यथा काष्ठेषु स्थितः अग्निः तमेव (iii) ………….. तथैव क्रोधरूपाग्निः अपि (iv) ………………. शरीराणि दहति।
मञ्जूषा- शत्रुः, जनानाम्, क्रोधः, दहति
Answer
Answer:
(i) शत्रुः
(ii) क्रोधः
(iii) दहति
(iv) जनानाम्
(च) मृगाः मृगैः सङ्गमनुव्रजन्ति,
गावश्च गोभिः तुरगास्तुरङ्गैः।
मूर्खाश्च मूखैः सुधियः सुधीभिः,
समान-शील-व्यसनेषु सख्यम्॥
भावार्थ:- अस्य श्लोकस्य भावोऽस्ति यत् संसारे सर्वे जीवाः समानः (i) …………… स्वभावयुक्तैः जीवैः सह मैत्री कुर्वन्ति। यथा मृगाः (ii) …………… सह गावः गोभिः सह अश्वाः (iii) ………… सह, मूर्खा, मूखैः सह, विद्वांस (iv) ……………. सह एव अनुगच्छन्ति।
मञ्जूषा- विद्वद्भिः, मृगैः, व्यवहारः, अश्वैः
Answer
Answer:
(i) व्यवहारः
(ii) मृगैः
(iii) अश्वैः
(iv) विद्वद्भिः
(छ) सेवितव्यो महावक्षः फलच्छायासमन्वितः।
यदि दैवात् फलं नास्ति छाया केन निवार्यते॥
भावार्थ:- संसारे सदैव फलैः छायया च (i) …………………. महान् वृक्षः (ii) …………….. भवति। यदि दुर्भाग्यवशात् तस्मिन् (iii) ……………. नापि भवेयुः तथापि तं (iv) ……………. दातुं कश्चिदपि रोढुं न शक्नोति।
मञ्जूषा- छायां, आश्रयितव्यः, युक्तः, फलानि
Answer
Answer: (i) युक्तः
(ii) आश्रयितव्यः
(iii) छायां
(iv) फलानि
(ज) अमन्त्रमक्षरं नास्ति, नास्ति मूलमनौषधम्।
अयोग्यः पुरुषः नास्ति योजकस्तत्र दुर्लभः ॥ 8 ॥
भावार्थ:- अस्मिन् संसारे किञ्चिद् (i) ………………… ज्ञानम् (अक्षरम्) न भवति। एवमेव औषधि गुण रहितं वृक्षाणां (ii) ………………. अपि न भवति। योग्यता रहितः जनः (iii) ……………. सह (iv) …………….. संसारे अतीव दुर्लभोभवति।
मञ्जूषा- मूलम्, योजकः, अमन्त्रम्, पुरुषः
Answer
Answer:
(i) अमन्त्रम्
(ii) मूलम्
(iii) पुरुषः
(iv) योजकः
(झ) संपत्तौ च विपत्तौ च महतामेकरूपता।
उदये सविता रक्तो रक्तोश्चास्तमये तथा॥
भावार्थ:- अस्य भावोऽस्ति यत् यथा सूर्यः (i) ……………….. समये रक्तः भवति तथैव सः अस्ते समये अपि (ii) …………….. भवति तथैव महान्तः (iii) ……………… सम्पत्ति आगते सति अपि शान्ताः भवति एवमेव (iv) …………….. आगते अपि ते शान्ताः एव तिष्ठन्ति।
मञ्जूषा- जनाः, विपत्तौ, उदये, रक्तः
Answer
Answer: (i) उदये
(ii) रक्तः
(iii) जनाः
(iv) विपत्तौ
(ञ) विचित्रे खलु संसारे नास्ति किञ्चिन्निरर्थकम्।
अश्वश्चेद् धावने वीरः भारस्य वहने खरः॥
भावार्थ:- अर्थात् अस्मिन् विचित्रे (i) …………… किञ्चिदपि वस्तु (ii) ……………… नास्ति। यतः यदा धावनस्य कार्यं भवति तदा (iii) ………………… प्रयोगः क्रियते परन्तु यदा भार वहनस्य (iv) ……………. क्रियते तदा खरः उपयोगी भवति।
मञ्जूषा- संसारे, अश्वस्य, कार्य, निरर्थकम्
Answer
Answer:
(i) संसारे
(ii) अश्वस्य
(iii) कार्य
(iv) निरर्थकम्
पर्यायपदानि-उचितं पर्याय मेलनं कुरुत
खण्ड ‘क’ – खण्ड ‘ख’
(क) प्रसीदति – दुखम् अनुभवति
(ख) ध्रुवं – अश्वाः
(ग) निमित्तः – जानाति
(घ) करी – कथितः
(ङ) वेत्ति – प्राप्यते
(च) अवसीदति – बुद्धिमान्
(छ) अपगमे – नमन्त्रम्
(ज) उदीरितः – मननयोग्य
(झ) गृह्यते – अग्निः
(ञ) हयाः – गर्दभः
(ट) नागाः – समाप्ते
(ठ) पण्डितः – विद्वांसः
(ड) वह्नि – भाग्यात्
(ढ) तुरगाः – गजाः
(ण) सुधियः – कारणम्
(त) सख्यम् – सूर्यः
(थ) व्यसनेषु – अश्वाः
(द) दैवात् – प्रसन्नः भवति
(ध) सविता – स्वभावे
(न) अमन्त्रम् – निश्चितम्
(प) खरः – मैत्री
(फ) मन्त्र – गजः
Answer
Answer:
खण्ड ‘क’ – खण्ड ‘ख’
(क) प्रसीदति – प्रसन्नः भवति
(ख) ध्रुवं – निश्चितम्
(ग) निमित्तः – कारणम्
(घ) करी – गजः
(ङ) वेत्ति – जानाति
(च) अवसीदति – दुखम् अनुभवति
(छ) अपगमे – समाप्ते
(ज) उदीरितः – कथितः
(झ) गृह्यते – प्राप्यते
(ञ) हयाः – अश्वाः
(ट) नागाः – गजाः
(ठ) पण्डितः – बुद्धिमान्
(ड) वह्नि – अग्निः
(ढ) तुरगाः – अश्वाः
(ण) सुधियः – विद्वांसः
(त) सख्यम् – मैत्री
(थ) व्यसनेषु – स्वभावे
(द) दैवात् – भाग्यात्
(ध) सविता – सूर्यः
(न) अमन्त्रम् – नमन्त्रम्
(प) खरः – गर्दभः
(फ) मन्त्र – मननयोग्य
विशेषण-विशेष्यपदानि योजयत
विशेषण पदानि – विशेष्य पदानि
(क) महान् – (i) शत्रुः
(ख) प्रथमः – (ii) शील
(ग) समान – (iii) पुस्तके
(घ) शोभने – (iv) पुरुषः
(ङ) लघुः – (v) रिपुः
(च) श्रेष्ठतम् – (vi) गीतानि
(छ) मधुराणि – (vii) कथा
Answer
Answer:
(क) महान् – (v) रिपुः
(ख) प्रथमः – (i) शत्रुः
(ग) समान – (ii) शील
(घ) शोभने – (iii) पुस्तके
(ङ) लघुः – (vii) कथा
(च) श्रेष्ठतम् – (iv) पुरुषः
(छ) मधुराणि – (vi) गीतानि
संस्कृतेन वाक्यप्रयोगं कुरुत

Answer
Answer:
(क) नीलः मेघः आकाशे सञ्चरति।
(ख) सुन्दरी बालिका गीतं गायति।
(ग) अतं मधुरं फलं खादामि।
(घ) रामः दशरथस्य प्रियः पुत्रः आसीत्।
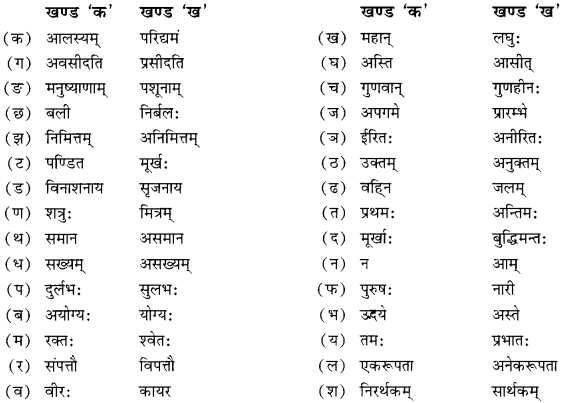
विपर्ययमेलनं कुरुत
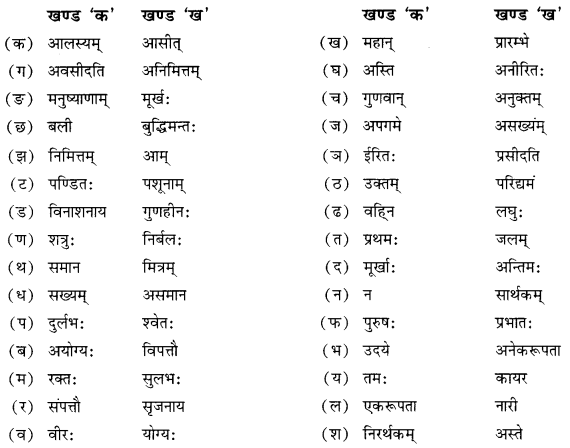
Answer
Answer:
We hope the given NCERT MCQ Questions for Class 10 Sanskrit Chapter 6 सुभाषितानि with Answers Pdf free download will help you. If you have any queries regarding CBSE Class 10 Sanskrit सुभाषितानि MCQs Multiple Choice Questions with Answers, drop a comment below and we will get back to you soon.