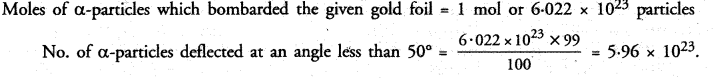NCERT Solutions for Class 9 Science Chapter 5 The Fundamental Unit of Life
These Solutions are part of NCERT Solutions for Class 9 Science. Here we have given NCERT Solutions for Class 9 Science Chapter 5 The Fundamental Unit of Life. LearnInsta.com provides you the Free PDF download of NCERT Solutions for Class 9 Science (Biology) Chapter 5 – The Fundamental Unit of Life solved by Expert Teachers as per NCERT (CBSE) Book guidelines. All Chapter 5 – The Fundamental Unit of Life Exercise Questions with Solutions to help you to revise complete Syllabus and Score More marks.
More Resources
- NCERT Solutions for Class 9 Science
- HOTS Questions for Class 9 Science
- Value Based Questions in Science for Class 9
- NCERT Exemplar Solutions for Class 9 Science
- Previous Year Question Papers for CBSE Class 9 Science
NCERT TEXT BOOK QUESTIONS
IN TEXT QUESTIONS
Question 1.
Who discovered cell, and how ?
Answer:
Robert Hooke (1665). He thinly pared a piece of cork and observed it under his self made primitive microscope. The scientist found that cork possesses a number of small box-like structures which he named cells (cellulae which later’abbreviated to cells). His work was published in the form of a book called Micrographia.
Question 2.
Why is cell called structural and functional unit of life ?
Answer:
Structural Unit: A living organism is made up of one or more cells. Therefore, cell is structural unit of life. Functional Unit. All life functions of an organism reside in its cells. Cells may also become specialised to perform specific functions like contraction in muscle cell or impulse transmission in nerve cell. Therefore, cells are functional units of life.
Question 3.
How do substances like CO2 and water move into and out of the cell ? Discuss. (CCE 2011)
Answer:
CO2 moves into and out of cells by diffusion while water does it through osmosis.
Diffusion. It is movement of particles of various substances from the region of their higher concentration to the region of their lower concentration,
(i) In a respiring cell, more CO2 is produced internally. As a result its internal concentration rises. As concentration of CO2 is lower in the outside medium, CO2 passes out from cell into external medium,
(ii) In photosynthetic cell, CO2 is being consumed in photosynthesis. Its intracellular concentration is lower than outside medium. Therefore, CO2 diffuses from outside to inside of the cell. Osmosis. It is movement of water from the region of its higher concentration (pure water or dilute solution) to the region of its lower concentration (strong solution) when the two are separated by a semipermeable membrane. Plasma membrane functions as a semipermeable membrane. Cell sap functions as strong solution. Therefore, external water enters the cell (endosmosis) till wall pressure counter-balances this tendency. If the external medium has a very strong solution, water would pass out from the cell into the external medium. The phenomenon is called exosmosis.
Question 4.
Why is plasma membrane called selectively permeable membrane ? (CCE 2012)
Answer:
Cell membrane is semipermeable membrane for water. It permits the entry of gases through diffusion. Ions, sugar, amino acids, etc. pass through the plasma membrane by an active process. Plasma membrane is impermeable to certain other materials. Therefore, it is selectively permeable.
Question 5.
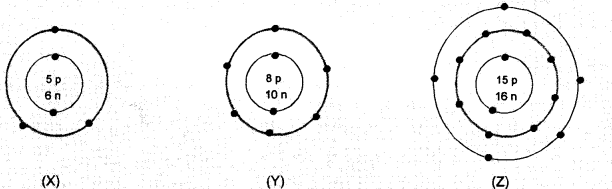
Fill in the gaps in the following table illustrating differences between prokaryotic and eukaryotic cells.

Answer:
2. (Left side), (i) Poorly defined due to absence of nuclear envelope (ii) Nucleoid.
4. (Right side). Membrane bound cell organelles are present.
Question 6.
Can you name the two organelles, we have studied that contain their own genetic material ?
Answer:
Yes. Mitochondria and plasdds.
Question 7.
If the organisation of a cell is destroyed due to some physical or chemical influence, what will happen ?
Answer:
Lysosome will burst to release digestive enzymes. Digestive enzymes will cause breakdown of various cellular components causing destruction of the cell.
Question 8.
Why are lysosomes known as suicide bags ? (CCE 2011, 2012, 2013)
Answer:
Lysosomes contain digestive enzymes against all types of organic materials. If their covering membrane breaks as it happens during injury to cell, the digestive enzymes will spill over the cell contents and digest the same. As lysosomes are organelles which on bursting can kill the cells possessing them, they are called suicide bags.
Question 9.
Where are proteins synthesised inside the cell ?
Answer:
Proteins are synthesized over the ribosomes.
NCERT Exercise
Question 1.
Make a comparison to write down ways in which plant cells are different from animal cells.
Answer:

Question 2.
How is prokaryotic cell different from eukaryotic cell ?
Answer:

Question 3.
What would happen if the plasma membrane ruptures or breaks down ?
Answer:
There will be spilling of cytoplasm and cell organelles, bursting of lysosomes and digestion of cellular contents.
Question 4.
What would happen to the life of a cell if there was no Golgi apparatus ? (CCE 2011)
Answer:
There would not be any lysosome for intracellular digestion and cleansing, no complexing of molecules, no excretion and no formation of new plasma membrane.
Question 5.
Which organelle is known as power house of the cell ? Why ? (CCE 2011, 2013)
Answer:
Mitochondrion is known as power house of the cell because it produces most of the molecules of ATP (adenosine triphosphate) which are required for providing energy for synthesis of new chemicals, mechanical and other cellular functions.
Question 6.
Where do the lipids and proteins constituting the cell membrane get synthesised ?
Answer:
Proteins are synthesised over ribosomes of RER while lipids are synthesised over SER.
Question 7.
How does Amoeba obtain food ?
Answer:
Plasma membrane of Amoeba is flexible. With its help, Amoeba engulfs food particle. The engulfed food particle passes into the body of Amoeba as a phagosome. Phagosome combines with lysosome to produce digestive or food vacuole. Digestion occurs in food vacuole. The digested food passes into surrounding cytoplasm. The undigested matter is thrown out of the cell in exocytosis.
Question 8.
What is osmosis ?
Answer:
Osmosis is diffusion of water from the region of its higher concentration (pure water or dilute solution) to the region of its lower concentration (strong solution) through a semipermeable membrane.
Question 9.
Carry out the following osmosis experiment.
Take four peeled potato halves and hollow each one out to make potato cups. One of these potato cups should be made from the boiled potato. Put each potato cup in the trough containing water. Now
(a) Keep cup A empty,
(b) Put one tea spoon sugar in cup B.
(c) Put one tea spoon of salt in cup C.
(d) Put one tea spoon sugar in boiled cup D.
Keep this set up for two hours. Then observe the four potato cups and answer the following :
(i) Explain why water gathers in the hollowed portion of B and C.
(ii) Why is potato A necessary for this experiment ?
(iii) Explain why water does not gather in the hollowed out portion of A and D.
Answer:
(i) Sugar and salt increases osmotic concentration which results in passage of water osmotically from the trough through the cells of potato B and C into its cavity.
(ii) Potato A functions as control experiment which indicates that the cavity of potato does not induce movement of water. Water does not gather in the hollowed out portion of A because it does not have a higher osmotic concentration than the cells of potato tuber.
(iii) Potato tuber D does not have living cells. Osmosis does not occur in dead cells. Therefore, despite presence of sugar in the cavity of D, no water passes from trough through dead potato cells into cavity of the tuber.
PRACTICAL BASED TWO MARKS QUESTIONS
Question 1.
Three students ‘A’, ‘B’ and ‘C were given five raisins each of equal mass. The raisins were soaked in distilled water at room temperature. ‘A’ soaked the raisins for 10 minutes, ‘B’ for overnight and ‘C for 60 minutes. They calculated the percentage of water absorbed by raisins. Now answer the following question :
(a) Name the student whose raisins will show the maximum percentage of water absorbed.
(b) Name the student whose raisins will show the minimum percentage of water absorbed. (CCE 2013)
Answer:
(a) Maximum Percentage ofWater Absorbed. Raisins of ‘B’ student.
(b) Minimum Percentage of Water Absorbed. Raisins of ‘A’ student.
Question 2.
A teacher soaked 10 g raisins in 35 ml of distilled water in a beaker A and a similar amount in beaker B. She maintained the temperature of beaker A at 20°C and beaker B at 40°C. After an hour, compare the percentage of water absorbed by raisins in beakers A and B. (CCE 2013, 2014)
Answer:
More water was absorbed in beaker B where temperature was 40°C. Permeability of cell membrane increases from 0°C to 40°C beyond which dénaturation sets in.
Question 3.
5g of raisins were placed in distilled water for 24 hours. The mass of water soaked raisins was found to be 7g. Calculate the percentage of water absorbed by raisins. (CCE 2013)
Answer:
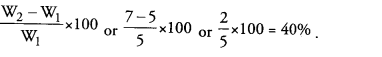
Question 4.
A student puts five raisins in two beakers A and B. Beaker A contained 50 ml of distilled water at room temperature and beaker B had 50 ml of ice cold water. After some time what will be the observation of the student ? State reason for this observation.
(CCE 2013, 2015)
Answer:
Raisins in beaker A swell up. Those of beaker B do not. Low temperature of B water reduced membrane permeability as well as kinetic energy of water for osmosis.
Question 5.
A student recorded a mass of dry raisins as 6.0 g and mass of raisins after soaking them in water for about four hours as 10.5 g. Calculate the percentage of water absorbed by raisins. (CCE 2013)
Answer:
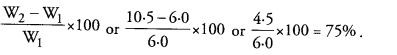
Question 6.
Write the mathematic equation used to determine the mass percentage of water absorbed by raisins.
Answer:
If initial weight is W1 and final weight after soaking is W2
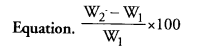
Question 7.
Ravi took weight of five dry raisins and five swollen raisins of approximately equal size. If the weight of dry raisins was 7 g and weight of swollen raisins is ‘X’ g, then
(i) Write the formula to calculate the percentage of water absorbed by the raisins and
(ii) If the value of ‘X’ is 10.5 g, then what will be the percentage of water absorbed by the raisins ? (CCE 2014)
Answer:
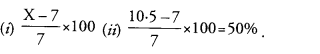
Question 8.
Write four main steps of the method involved in an experiment “On determination of the percentage of water absorbed by raisins in the laboratory”. (CCE 2014)
Answer:
(i) Selection of raisins with intact stalks and their weight as W1
(ii) Soaking the raisins in water at room temperature for at least 1-2 hours
(iii) Taking out wet raisins, wiping out water with blotting paper and their final weight as W2
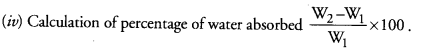
Question 9.
In the experiment of determining the percentage of water absorbed by raisins, two students Samya and Zahira used the following formula respectively
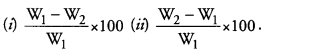
Which student used wrong formula ? What are W1 and W2 in the correct formula ? (CCE 2014)
Answer:
Student (i) used the wrong formula.
W1 is initial weight of dry raisins. W2 is final weight of swollen raisins.
Question 10.
(a) A student recorded the mass of dry raisins as 4.0 g and mass of raisins after soaking as 7.0 g. Calculate the percentage of water absorbed by raisins. (CCE 2014, 2016)
(b) Mention one application of the phenomenon of osmosis in plants. (CCE 2016)
Answer:
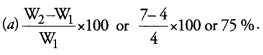
(b) Application of Osmosis. Turgidity of cells, root absorption.
Question 11.
(a) Ram while doing an experiment to find out the percentage of water absorbed by raisins measured the mass of dry raisins as 50 g. He soaked the raisins in water for four hours and again measured the mass as 80 g. Calculate the percentage of water absorbed by the raisins.
(b) He then placed raisins in concentrated salt solution. What will he observe ? (CCE 2015)
Answer:
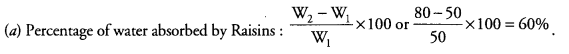
(b) After Placing in Concentrated Salt Solution. Raisins will lose water and shrink to the maximum.
Question 12.
A student recorded the mass of dry raisins as 6.0 g and mass of raisins after soaking them in water for about four hours as 10.5 g. Calculate the percentage of water absorbed by raisins. Why do raisins get swelled up ? (CCE 2015)
Answer:
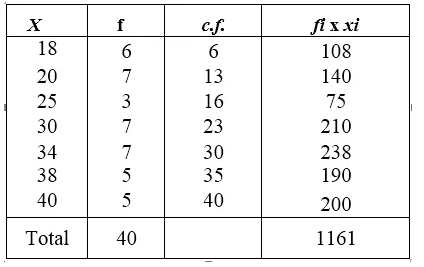
![]()
The raisins swell up due to absorption of water through osmosis.
Question 13.
A student recorded the following observations in an experiment for finding the percentage of water absorbed by raisins.
(i) Mass of water taken in beaker – 50 g
(ii) Mass of dry raisins before soaking water – 20 g
(iii) Mass of raisins after soaking water – 30 g.
(iv) Mass of remaining water in beaker after experiment – 40 g
Calculate the percentage of water absorbed by raisins. ( CCE 2015)
Answer:
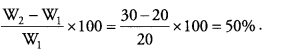
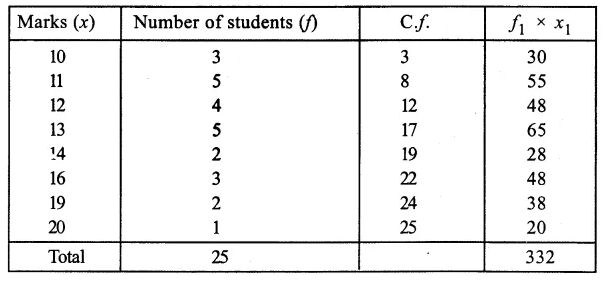
Question 14.
If ‘X’ is the initial mass of the raisins and ‘Y’ is the final mass of raisins after soaking in water, calculate the percentage of water absorbed by raisins. Name the process due to which raisins absorb water. (CBSE 2015, 2016)
Answer:
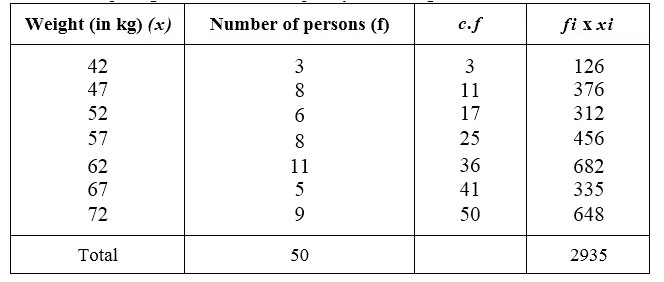
![]()
Question 15.
A group of students selected 10 raisins with stalks and weighed them using digital balance. Then, they soaked them for a few hours. The weight of the swollen raisins was 9.2 g which was 4.6 g more than weight of dry raisins. Calculate the water imbibed by the raisins. ( CCE 2015)
Answer:
![]()
Question 16.
In the experiment “To determine the mass percentage of water imbibed by raisins”, the raisins absorb water when kept in water for 5-6 hours. Why does water absorption take place ? What is the phenomenon called ? (CCE 2015, 2016)
Answer:
Water moves into raisins due to endosmosis. Endosmosis is entry of water into a system, cell or organ due to presence of hypertonic solution in it and its separation from pure water or dilute solution by a semipermeable membrance. Skin of raisins function as semipermeable membrane. There is high concentration of sugar inside them. Therefore, external water passes into raisins and cause their swelling.
Question 17.
Before placing the raisins in water, the raisins weighed 10 g. The raisins were taken out of water, wiped well and then the weight was found to be 12.5 g. Determine the percentage of water absorbed by raisins. Define the process due to which raisins absorb water. (CCE 2016)
Answer:
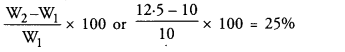
Osmosis: Osmosis is the diffusion of water or solvent across a semipermeable membrane (which does not allow passage of solutes) from a region of its higher concentration to the region of its lower concentration. Plasma membrane functions as semipermeable membrane. There will be osmotic entry of water into cell if the external solution is pure water or dilute as compared to cell sap.
Question 18.
(a) In the experiment to determine the percentage of water absorbed by raisins, the raisins are wiped before weighing. Why ?
(b) While preparing a temporary stained mount of onion peel, Veena added a drop of glycerine. Why ? ( CCE 2016)
Answer:
(a) To remove unabsorbed water sticking to the surface of raisins.
(b) To prevent drying of onion peel.
Question 19.
A student took x gram water in a beaker and dipped p gram raisins in it. After keeping raisins in water for 2 hours, he measured the mass of soaked raisins as q grams. He also measured the mass of water left in the beaker which was y grams. On the
Answer:
![]()
SELECTION TYPE QUESTIONS
basis of his observations write correct formula to find the percentage of water absorbed by raisins. Mention the process due to which weight of raisins increased. (CCE 2016)
Alternate Response Type Questions
(True/False, Right/Wrong, Yes/No)
Question 1.
Cork comes from bark.
Question 2.
Robert Brown discovered protoplasm in 1831.
Question 3.
Amoeba has an everchanging shape.
Question 4.
Movement of a substance from the area of low concentration to an area of high concentration is called diffusion.
Question 5.
A dilute soludon is called hypertonic solution.
Question 6.
Lysosomes keep the cells clean by digesting foreign materials and worn out cell organelles.
Question 7.
SER detoxifies many poisons and drugs.
Question 8.
Central vacuole occupies 10-20% of cell volume.
Matching Type Questions :
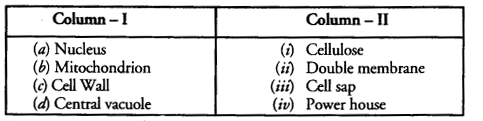
Question 9.
Match the contents of the columns I and II (single matching)
Question 10.
Match the contents of columns I, II and III (double matching)

Question 11.
Which type of metabolism, anabolism (A) and catabolism (C) are performed by the following organelles (key or check list items) :
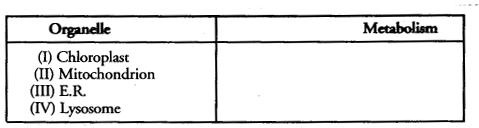
Question 12.
Match the stimulus with Appropriate Response.
Fill In the Blanks
Question 13. Cristae create a large surface area for …………… generating reactions.
Question 14. Plant cell wall is mainly composed of ……………..
Question 15. Same organelles perform …………… function in all organisms.
Question 16. Cell theory was proposed by …………. and ……………. .
Question 17. Cells were discovered by Robert Hooke in ………….. .
Answers:

SOME TYPICAL QUESTIONS
Question 1.
What is the junctional unit of life ? Define it.
Answer:
Cell is the functional unit of life. It can be defined as a tiny mass of protoplasm covered by plasma membrane which is capable of performing all functions of life.
Question 2.
What is the difference between plasma membrane and cell wall i Give the functions of each one. (CCE 2011)
Answer:
Plasma membrane is an elastic living membrane made up of lipids and proteins, whereas cell wall is a rigid non-living covering made up of cellulose.
Function of Plasma membrane. It acts as a selectively semipermeable membrane which allows only selective substances to pass through it.
Function of Cell Wall. It provides rigidity and protection to cell.
Question 3.
Differentiate between chromatin and chromosome.
Answer:
Chromatin. It is the nucleoprotein fine fibrous mass which stains strongly with basic dyes and is present as a network inside the nucleus.
Chromosome. Rod-like, stainable, condensed chromatin unit, visible at cell division and containing hereditary information in the form of genes.
Question 4.
Differentiate between RER and SER.
Answer:

Question 5.
Which type of ribosomes are found in prokaryotes and eukaryotes ?
Answer:
Prokaryotes have 70 S ribosomes and eukaryotes have 80 S ribosomes.
Question 6.
Which structure is called little nucleus ?
Answer:
Nucleolus.
Question 7.
Why is nucleus called director of the cell ?
Answer:
Nucleus controls and coordinates all the metabolic functions of the cell.
NCERT Solutions for Class 9 Science Chapter 5 The Fundamental Unit of Life
Hope given NCERT Solutions for Class 9 Science Chapter 5 are helpful to complete your science homework.
If you have any doubts, please comment below. Learn Insta try to provide online science tutoring for you.




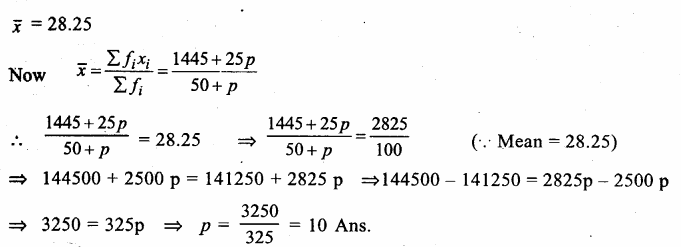
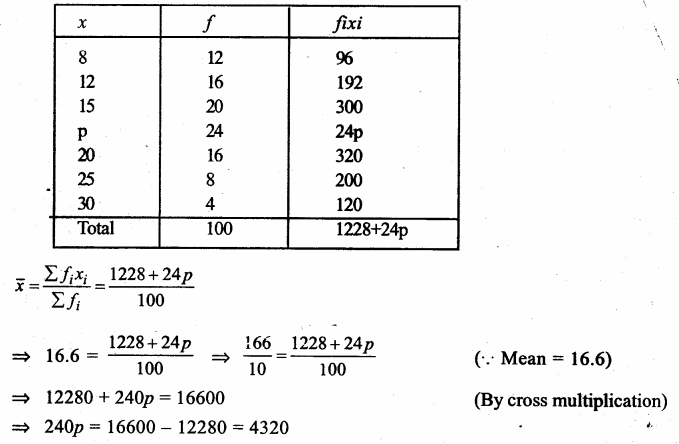
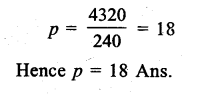

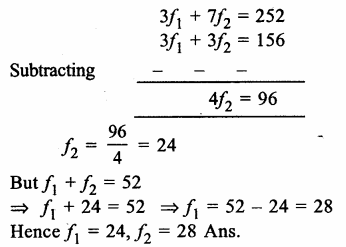
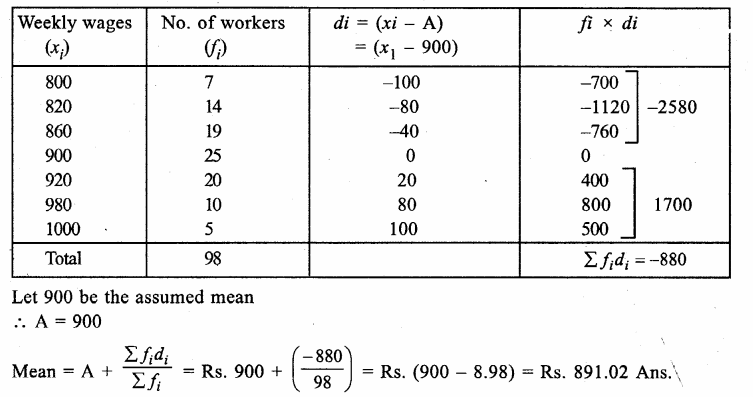
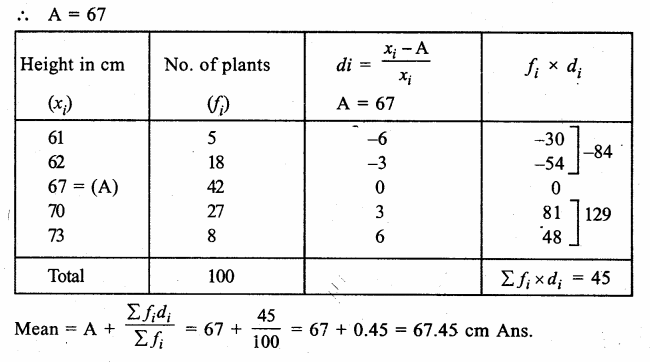
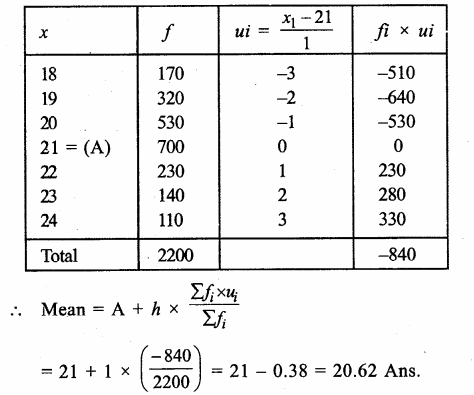

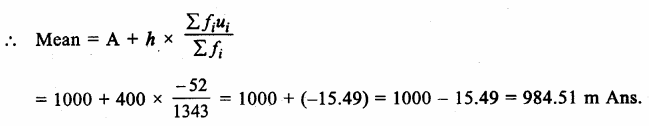
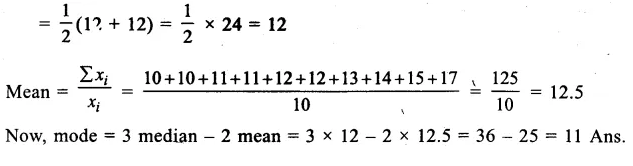
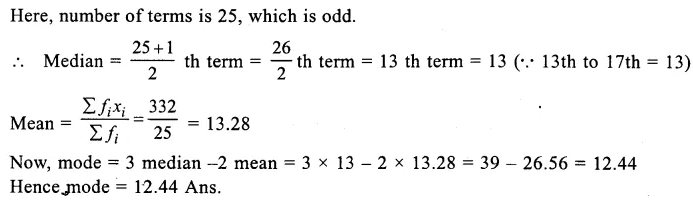

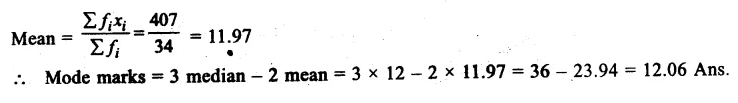
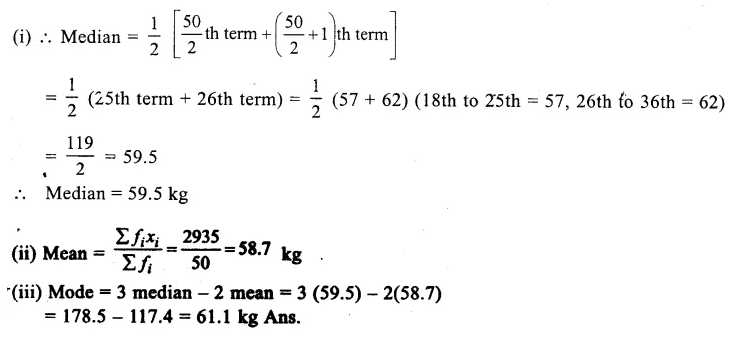

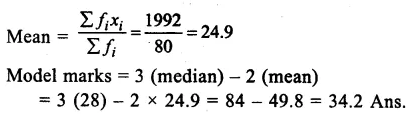


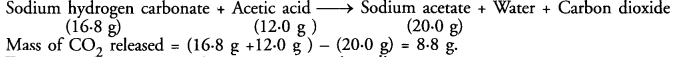

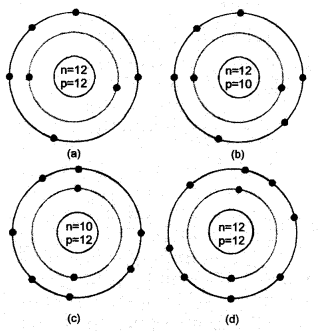
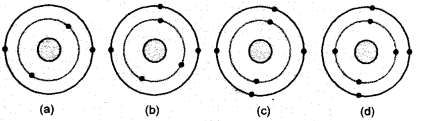
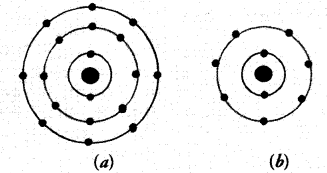 Answer:
Answer: