NCERT Exemplar Solutions for Class 10 Science Chapter 4 Carbon and Its Compounds
NCERT Exemplar Solutions for Class 10 Science Chapter 4 Multiple Choice Questions
Question 1.
Carbon exists in the atmosphere in the form of
(a) carbon monoxide only
(b) carbon monoxide and carbon dioxide in traces
(c) carbon dioxide only
(d) coal gas.
Answer:
(b). Both CO and CO2 gases are present in traces in the atmosphere.
More Resources
- NCERT Exemplar Solutions for Class 10 Science
- NCERT Solutions for Class 10 Science
- Value Based Questions in Science for Class 10
- HOTS Questions for Class 10 Science
- Previous Year Question Papers for CBSE Class 10 Science
Question 2.
Which of the following statements are usually correct for carbon compounds ? These
(i) are good conductors of heat and electricity
(ii) are poor conductors of electricity
(iii) have strong forces of attraction between their molecules
(iv) do not have strong forces of attraction between their molecules
(a) (i) and (iii)
(b) (ii) and (iii)
(c) (i) and (iv)
(d) (ii) and (iv)
Answer:
(d). Carbon compounds are of covalent nature. That is why these are poor conductors of electricity and do not have strong forces of attraction.
Question 3.
A molecule of ammonia (NH3) has
(a) only single bonds
(b) only double bonds
(c) only triple bonds
(d) two double bonds and two single bonds.
Answer:
(a). All bonds are single covalent in nature.
Question 4.
Buckminster fullerene is an allotropie form of
(a) nitrogen
(b) sulphur
(c) carbon
(d) tin
Answer:
(c).
Question 5.
Which of the following are correct chain isomers of butane ?
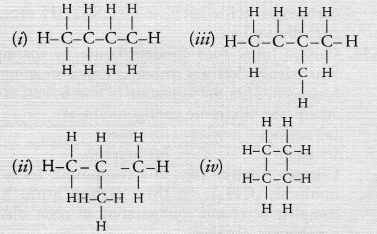
(a) (i) and (iii)
(b) (ii) and (iv)
(c) (i) and (ii)
(d) (iii) and (iv)
Answer:
(c). Isomer (i) has a straight chain while (ii) is a branched chain isomer.
Question 6.
![]()
In the above reaction, alkaline KMnO4 acts as
(a) reducing agent
(b) oxidising agent
(c) catalyst
(d) dehydrating agent.
Answer:
(b). Alkaline KMnO4 is also known as Baeyer’s reagent. It acts as an oxidising agent.
Question 7.
Oils on treating with hydrogen in the presence of palladium or nickel catalyst form fats. This is an example of
(a) Addition reaction
(b) Substitution reaction
(c) Displacement reaction
(d) Oxidation reaction
Answer:
(a). Oils are unsaturated in nature. By the addition of hydrogen, they become saturated.
Question 8.
In which of the following compounds, —OH is the functional group ?
(a) Butanone
(b) Butanol
(c) Butanoic acid
(d) Butanal
Answer:
(b). —OH is the functional group in butanol which is an alcohol.
Question 9.
The soap molecule has a
(a) hydrophilic head and a hydrophobic tail
(b) hydrophobic head and a hydrophilic tail
(c) hydrophobic head and a hydrophobic tail
(d) hydrophilic head and a hydrophilic tail
Answer:
(a). ‘Head’ is a polar group and is attacked towards H2O molecules of water. ‘Tail’ is a long chain of hydrocarbons and is water repellent.
Question 10.
Which of the following is the correct representation of electron dot structure of nitrogen ?
![]()
Answer:
(d). Both the N atoms have a complete octet after electron sharing.
Question 11.
Structural formula of ethyne is

Answer:
(a).
Question 12.
Identify the unsaturated compounds from the following
(i) Propane
(ii) Propene
(iii) Propyne
(iv) Chloropropane
(a) (i) and (ii)
(b) (ii) and (iv)
(c) (iii) and (iv)
(d) (ii) and (iii)
Answer:
(d). Both propene and propyne are unsaturated hydrocarbons.
Question 13.
Chlorine reacts with saturated hydrocarbons at room temperature in the
(a) absence of sunlight
(b) presence of sunlight
(c) presence of water
(d) presence of hydrochloric acid
Answer:
(b). These reactions are called photochemical reactions.
Question 14.
In the soap micelles
(a) the ionic end of soap is on the surface of the cluster while the carbon chain is in the interior of the cluster.
(b) ionic end of soap is in the interior of the cluster and the carbon chain is out of the cluster,
(c) both ionic end and carbon chain are in the interior of the cluster
(d) both ionic end and carbon chain are on the exterior of the cluster
Answer:
(a).
Question 15.
Pentane has the molecular formula C5H12. h has
(a) 8 covalent bonds
(b) 10 covalent bonds
(c) 16 covalent bonds
(d) 14 covalent bonds
Answer:
(c).
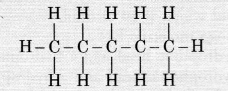
Question 16.
Structural formula of benzene is
Answer:
(c). The carbon atom skeleton in benzene has alternate single and double bonds
Question 17.
Ethanol reacts with sodium and forms two products. These are :
(a) sodium ethanoate and hydrogen
(b) sodium ethanoate and oxygen
(c) sodium ethoxide and hydrogen
(d) sodium ethoxide and oxygen
Answer:
(c).
![]()
Question 18.
The correct structural formula of butanoic acid is
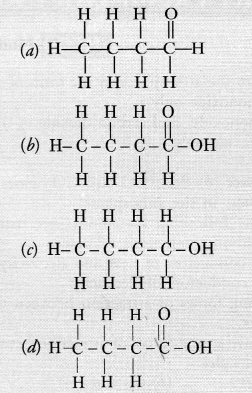
Answer:
(d).
Question 19.
Vinegar is a solution of
(a) 30% — 40% acetic acid in alcohol
(b) 5% — 8% acetic acid in alcohol
(c) 5% – 8% acetic acid in water
(d) 15% – 20% acetic acid in water
Answer:
(c).
Question 20.
Mineral acids are stronger acids than carboxylic acids because
(i) mineral acids are completely ionised
(ii) carboxylic acids are completely ionised
(iii) mineral acids are partially ionised
(iv) carboxylic acids are partially ionised
(a) (i) and (iv)
(b) (ii) and (iii)
(c) (i) and (ii)
(d) (iii) and (iv)
Answer:
(a). Mineral acids like HCl are completely ionised in solvent like water whereas carboxylic acids such as CH3COOH are only partially ionised.
Question 21.
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms e.g. hydrogen. After the formation of four bonds, carbon attains the electronic configuration of
(a) helium
(b) neon
(c) argon
(d) krypton
Answer:
(b). The compound formed is methane (CH4). In this, carbon atom has a complete octet and configuration of neon which is a noble gas element.
Question 22.
The correct electron dot-structure of a water molecule is
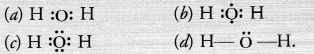
Answer:
(c).
Question 23.
Which of the following is not a straight chain hydrocarbon ?
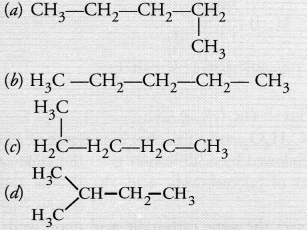
Answer:
(d). Please note that the continuous chains of carbon atoms whether straight or bent are not branched in nature.
Question 24.
Which among the following are unsaturated hydrocarbons ?
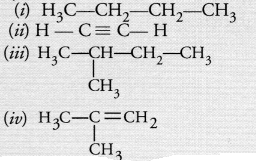
(a) (i) and (iii)
(b) (ii) and (iii)
(c) (ii) and (iv)
(d) (iii) and (iv).
Answer:
(c). Both (ii) and (iv) are unsaturated in nature.
Question 25.
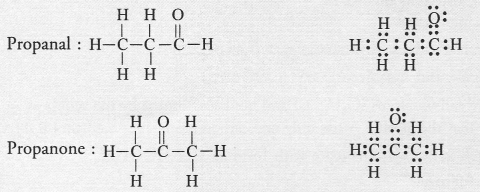
The IUPAC name of the compound
CH3—CH2—CHO is
(a) Propanal
(b) Propanone
(c) Ethanol
(d) Ethanal
Answer:
(a).
Question 26.
The heteroatoms present in CH3—O—CH2—CH2 (Br) are
(i) oxygen
(ii) carbon
(iii) hydrogen
(iv) bromine
(a) (i) and (ii)
(b) (ii) and (iii)
(c) (iii) and (iv)
(d) (i) and (iv)
Answer:
(d). Both oxygen (O) and bromine (Br) are heteroatoms. Please remember that apart from C and H atoms, all other atoms present in an organic compound are hetero atoms.
Question 27.
Which of the following does not belong to the same homologous series ?
(a) CH4
(b) C2H6
(c) C3H8
(d) C3H6.
Answer:
(d). It is an alkene while the rest are all alkane molecules in nature.
Question 28.
The first member of alkene family is
(a) ethyne
(b) ethene
(c) propyne
(d) ethane
Answer:
(b).
Question 29.
Which of the following represents saponification
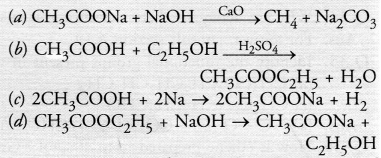
Answer:
(d). This is an example of saponification reaction. All other options are not correct.
NCERT Exemplar Solutions for Class 10 Science Chapter 4 Short Answer Questions
Question 30.
Draw the electron dot structure of ethyne and also draw its structural formula
Answer:
![]()
Question 31.
Write the names of the following compounds :

Answer:
(a) Pentanoic acid
(b) But-l-yne
(c) Heptanal
(d) Pentan-l-ol
Question 32.
Identify and name the functional groups present in the following compounds.
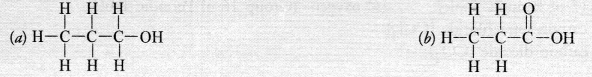

Answer:

Question 33.
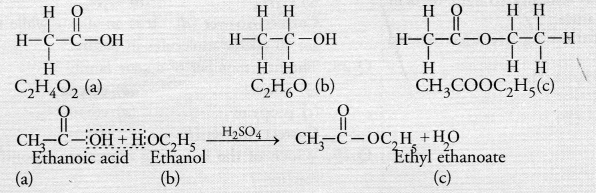
A compound X is formed by the reaction of a carboxylic acid C2H4O2 and an alcohol in presence of a few drops of H2SO4. The alcohol on oxidation with alkaline KMnO4 followed by acidification gives the same carboxylic acid as used in this reaction. Give the names and structures of (a) carboxylic acid,
(b) alcohol and
(c) the compound X. Also write the reaction. (CBSE Sample Paper 2017)
Answer:
The available information suggests that the alcohol which gives the same carboxylic acid upon oxidation has two carbon atoms. It is, therefore ethanol (C2H5OH). The structures of the different compounds are :
Question 34.
Why are detergents better cleansing agents than soaps ? Explain.
Answer:
A substance capable of removing grease and dirt from any fabric or body is called detergent. The detergents are of two types Le. soapy and non-soapy detergents. The soapy detergents are soaps whereas the non-soapy detergents are synthetic detergents or simply detergents. Although both are cleansing agents, they differ in chemical composition. In the present chapter, we shall briefly discuss the composition and cleansing action of soaps and synthetic detergents.
Soaps are the sodium and potassium salts of long chain fatty acids with general formula RCOONa or RCOOK. The acids present have the formula RCOOH where R may have following values.
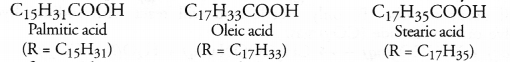
These fatty acids exist as triesters of glycerol which is a trihydric alcohol. The triesters are also called triglycerides or simply glycerides and are the constituents of edible oils and fats. These are of animal and vegetable origin e.g. castor oil, linseed oil or soyabean oil. Chemically the triglycerides are formed as a result of esterification reaction.
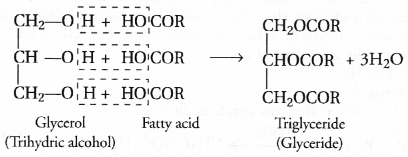
Question 35.
Identify the functional groups present in the following compounds
![]()
Answer:
(a) >C = O
(b) —COOH
(c) —CHO
(d) —OH.
Question 36.
How is ethene prepared from ethanol ? Give the reaction involved in it.
Answer:
From ethanol: This method involves the slow oxidation of a dilute solution of ethanol (10-15 per cent) by oxygen present in air in the presence of an enzyme acetobactor.
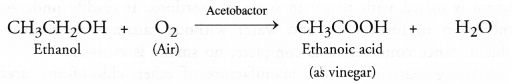
The acid obtained is in the form of dilute solution called vinegar.
We have also studied under ethanol that it gets oxidised to ethanoic acid in the presence of dilute solution of alkaline KMnO4 or acidified K2Cr2O7.
Question 37.
Intake of small quantity of methanol can be lethal. Comment.
Answer:
From methanol: These days ethanoic acid is manufactured by the reaction between methanol and carbon monoxide in the presence of iodine-rhodium (I2 — Rh) catalyst mixture.

Question 38.
A gas is evolved when ethanol reacts with sodium. Name the gas evolved and also write the balanced chemical equation for the reaction involved.
Answer:
The gas evolved is hydrogen. The balanced chemical equation for the reactibn is :
![]()
Question 39.
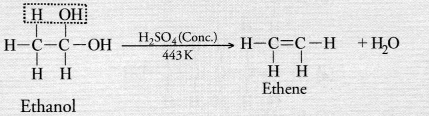
Ethene is formed when ethanol at 443 K is heated with excess of concentrated sulphuric acid. What is the role of sulphuric acid in this reaction ? Write the balanced chemical equation of this reaction.
Answer:
Concentrated sulphuric acid acts as a dehydrating agent in the reaction. Ethene is formed as the
Question 40.
Carbon, Group (14) element in the Periodic Table, is known to form compounds with many elements. Write an example of a compound formed with
(a) chlorine (Group 17 of Periodic Table)
(b) oxygen (Group 16 of Periodic Table)
Answer:
(a) The compound is carbon tetrachloride (CCl4)
(b) The compound is carbon dioxide (CO2)
Question 41.
In electron dot structure, the valence shell electrons are represented by crosses or dots.
(a) The atomic number of chlorine is 17. Write its electronic configuration
(b) Draw the electron dot structure of chlorine molecule.
Answer:
(a) 2, 8, 7
![]()
Question 42.
Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon. Compare the ability of catenation of the two elements. Give reasons.
Answer:
The tendency to show catenation is very small in case of Si as compared to C although both the elements belong to the same group (14). This is because of greater atomic size of Si atom (118 pm) than that of carbon (77 pm). As a result, the strength of Si—Si bond is less as compared to that of C—C bond. This means that lesser number of Si atoms can be linked to each other by covalent bonds as compared to carbon atoms.
Question 43.
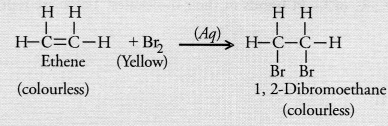
Unsaturated hydrocarbons contain multiple bonds between the two C-atoms and show addition reactions. Give the test to distinguish ethane from ethene.
Answer:
Distinction between ethane and ethene can be done with the help of bromine water test. Whereas ethene dicolourises the yellow colour of bromine water, ethane does not.
Question 44.
Match the reactions given in Column (A) with the names given in column (B).
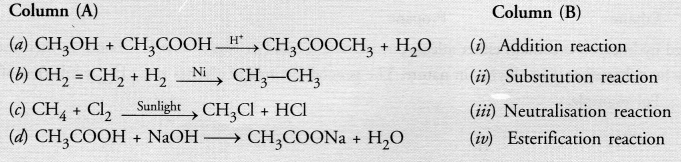
Answer:
(a)—(iv) ;
(b)—(i) ;
(c)—(ii) ;
(d)—(iii)
Question 45.
Write the structural formulae of all the isomers of hexane.
Answer:
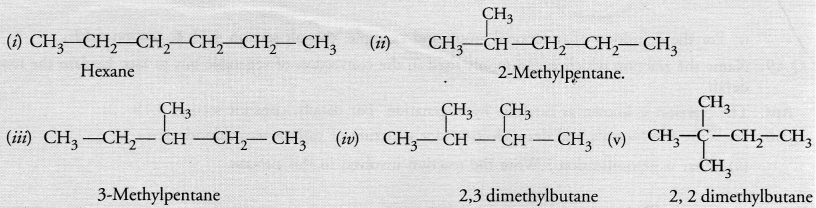
Question 46.
What is the role of metal or reagents written on arrows in the given chemical reactions?
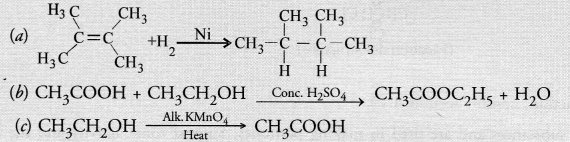
Answer:
(a) Nickel (Ni) acts as a hydrogenation catalyst for the reaction
(b) Cone. H2SO4 removes a molecule of H2O from the reaction mixture and acts as a dehydrating agent.
(c) Alkaline KMnO4 acts as an oxidising agent and oxidises ethanol to ethanoic acid.
NCERT Exemplar Solutions for Class 10 Science Chapter 4Long Answer Questions
Question 47.
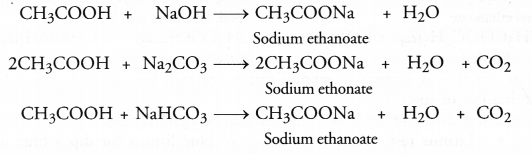
A salt X is formed and a gas is evolved when ethanoic acid reacts with sodium hydrogen carbonate. Name the salt X and the gas evolved. Describe an activity and draw the diagram of the apparatus to prove that the evolved gas is the one which you have named. Also, write chemical equation of the reaction involved.
Answer:
The salt X formed in the reaction is sodium ethanoate. The gas evolved is carbon dioxide gas. For the activity and chemical reaction,
Question 48.
(a) What are hydrocarbons ? Give examples.
(b) Give the structural differences between saturated and unsaturated hydrocarbons with two examples of each.
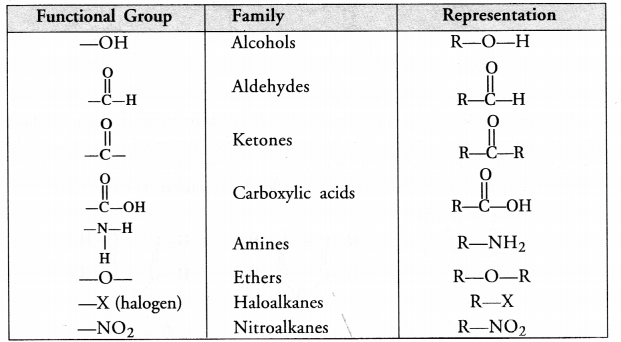
(c) What is a functional group ? Give examples of four different functional groups.
Answer:
(a) Hydrocarbons are the organic compounds containing only carbon and hydrogen atoms as their constituents. These may be alkanes, alkenes and alkynes.
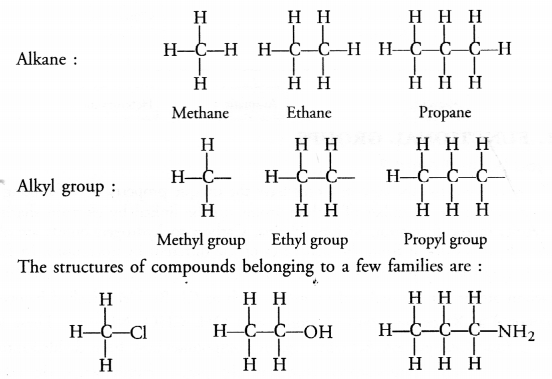
(b) Saturated hydrocarbons or alkanes contain either C—C or C—H bonds in their molecules. These are represented by the general formula CnH2n+2 For example,
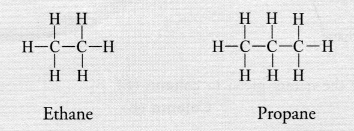
Unsaturated hydrocarbons contain either atleast one >C = C< bond or triple —C ≡ C— bond in their molecules. These may be either alkenes or alkynes in nature. The general formula of alkenes is CnH2n while that of alkynes is CnH2n-2 for example,
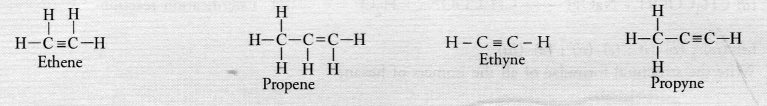
(c) For the definition of functional group and example,
Question 49.
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction in detail.
Answer:
The reaction is known as catalytic hydrogenation. For details, constult section 4.15.
Question 50.
(a) Write the formula and draw electron dot structure of carbon tetrachloride.
(b) What is saponification ? Write the reaction involved in this process.
Answer:

(b) For saponification reaction,

The reaction is known as saponification reaction because it is the basis for the formation of soap.
Question 51.
Esters are sweet-smelling substances and are used in making perfumes. Suggest some activity and the reaction involved for the preparation of an ester with well labelled diagram.
Answer:
Esters as pointed, are pleasant smelling compounds. These are therefore, commonly used as flavouring agents and also in perfumes. When an ester is reacted with water in the presence of a dilute acid like dilute HCl, acid and alcohol are formed as the product. The reaction is called ester hydrolysis.
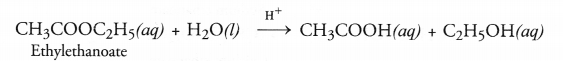
Ester hydrolysis is the reverse of esterification reaction.
When an ester is reacted with an aqueous solution of base like NaOH or KOH, the product is an alcohol and salt of the acid. For example,
The reaction is known as saponification reaction because it is the basis for the formation of soap.
Question 52.
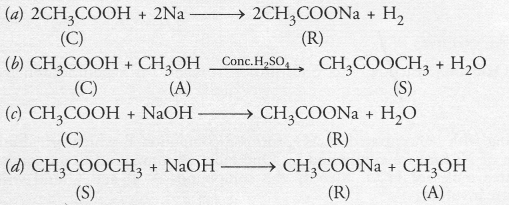
A compound C (molecular formula, C2H4O2) reacts with Na-metal to form a compound R and evolves a gas which burns with a pop sound. Compound C on treatment with an alcohol A in presence of an acid forms a sweet smelling compound S (molecular formula, C3H6O2). On addition of NaOH to C, it also gives R and water. S on treatment with NaOH solution gives back R and A.
Identify C, R, A, S and write down the reactions involved.
Answer:
From the available information, it is evident that
- Compound C with molecular formula C2H4O2 is ethanoic acid (CH3COOH)
- The compound R is sodium ethanoate and has formula CH3COONa
- Since the compound S has only three carbon atoms (C3H6O2) and has been formed by the action of an alcohol on compound C (C2H4O2), this means that the alcohol A has only one carbon atom. It is methanol (CH3OH).
- The compound S with a sweet smell is methyl ethanoate with formula CH3COOCH3.
The chemical reactions involved are as follows :
Question 53.
Look at given figure and answer the following questions :
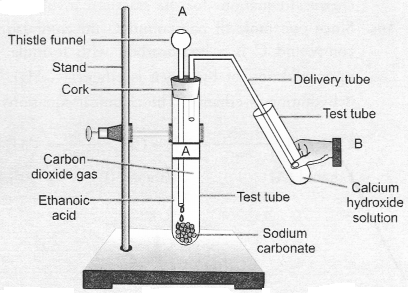
(a) What change would you observe in the calcium hydroxide solution taken in tube B ?
(b) Write the reaction involved in test tubes A and B respectively.
(c) If ethanol is used instead of ethanoic acid, would you expect the same change ?
(d) How can a solution of lime water be prepared in the laboratory ?
Answer:
(a) It would become milky.
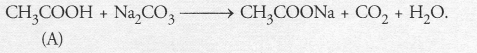
In test tube B, calcium hydroxide reacts with carbon dioxide to form calcium carbonate which is milky in colour.
![]()
(c) No, it would be different. No chemical reaction is possible between ethanol and sodium carbonate.
(d) Lime w’ater is prepared by keeping suspension of calcium hydroxide overnight in a beaker. The solution is decanted and is transferred to another beaker. It contains traces of calcium hydroxide and is called lime water.
Question 54.
How would you bring about the following conversions ? Name the process and write the reaction involved.
(a) ethanol to ethene.
(b) Methanol to Ethanoic acid. Write the reactions.
Answer:
(a) From ethanol: This method involves the slow oxidation of a dilute solution of ethanol (10-15 per cent) by oxygen present in air in the presence of an enzyme acetobactor.
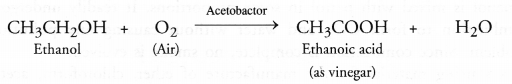
The acid obtained is in the form of dilute solution called vinegar.
We have also studied under ethanol that it gets oxidised to ethanoic acid in the presence of dilute solution of alkaline KMnO4 or acidified K2Cr2O7.
(b) From methanol: These days ethanoic acid is manufactured by the reaction between methanol and carbon monoxide in the presence of iodine-rhodium (I2 — Rh) catalyst mixture.

Question 55.
Draw the possible isomers of the compound with molecular formula C3H6O and also give their electron dot structures.
Answer:
Two isomers are possible for the molecular formula C3H6O. These are known as functional isomers.
Question 56.
Explain the given reactions with the examples :
(a) Hydrogenation reaction
(b) Oxidation reaction
(c) Substitution reaction
(d) Saponification reaction
(e) Combustion reaction
Answer:
Hydrogenation of oils : The reaction is extremely useful in the hydrogenation of vegetable oils also called edible oils e.g. ground nut oil, cotton seed oil etc. These are also called cooking oils and are unsaturated in the sense that their molecules contain atleast one C=C bond in their structures. Upon passing hydrogen gas through oil in the presence of nickel catalyst, the double bond changes to single bond. As a result, the unsaturated oil changes to solid fat which is of saturated nature. Vegetable ghees such as Dalda, are of saturated nature and are formed by catalytic hydrogenation reaction.
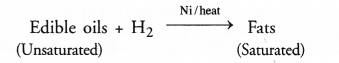
Oxidation reaction :
- Loss of hydrogen is known as oxidation.
- Gain of oxygen is known as oxidation.
Therefore, it is an oxidation reaction.
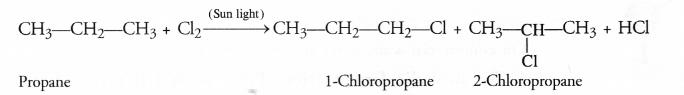
Substitution Reactions : Substitution reactions are also called replacement reactions and you are quite familiar with these. In organic compounds, particularly the saturated hydrocarbons (or alkanes), these reactions are very common. One or more hydrogen atoms in the molecule of alkane such methane get substituted by chlorine atoms when the reaction is carried with chlorine in the presence of ultraviolet sun light.
Combustion reaction :
We have seen that all combustion reactions are basically oxidation reactions carried in the presence of air or oxygen. It is not necessary that the reactants may burn during combustion.
- Blue flame signifies complete combustion of the fuel.
- Yellow sooty flame signifies incomplete combustion of the fuel.
Question 57.
An organic compound A on heating with concentrated H2SO4 forms a compound B which on addition of one mole of hydrogen in presence of Ni forms a compound C. One mole of compound C on combustion forms two moles of CO2 and three moles of H2O. Identify the compounds A, B and C and write the chemical equations for the reactions involved.
Answer:
Since one mole of compound C on combustion forms two moles of CO2 and three moles of H2O the compound C is a hydrocarbon with formula C2H6. It is ethane. The compound B which forms C2H6 upon addition of hydrogen is ethene (C2H4). The organic compound A which forms ethene upon acidic dehydration is ethanol. The chemical equations for the reactions involved are :
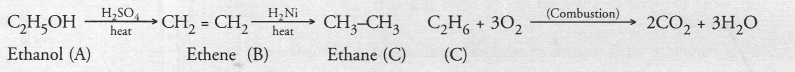
Hope given NCERT Exemplar Solutions for Class 10 Science Chapter 4 Carbon and Its Compounds helpful to you. If you have any doubts, please comment below. We try to provide online math tutoring for you.