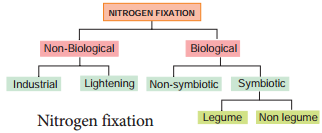
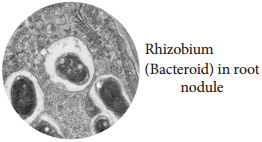
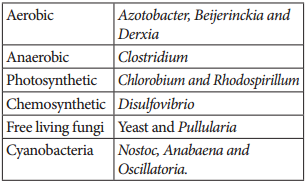
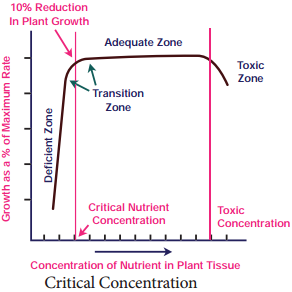
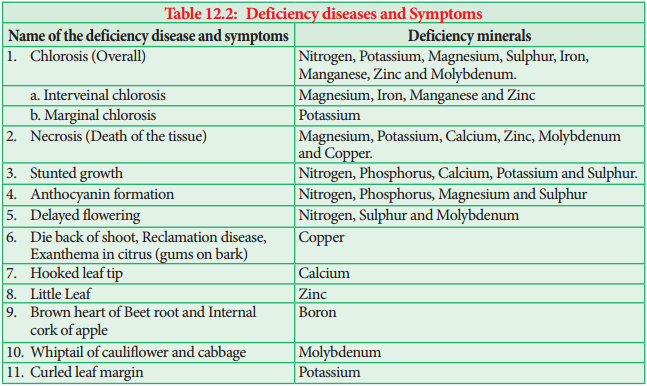
Learninsta presents the core concepts of Biology with high-quality research papers and topical review articles.
Difference Between Hydroponics and Aeroponics
1. Hydroponics or Soilless culture:
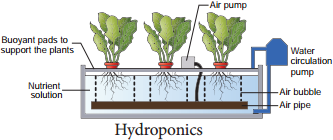
Von Sachs developed a method of growing plants in nutrient solution. The commonly used nutrient solutions are Knop solution (1865) and Arnon and Hoagland Solution (1940). Later the term Hydroponics was coined by Goerick (1940) and he also introduced commercial techniques for hydroponics. In hydroponics roots are immersed in the solution containing nutrients and air is supplied with help of tube (Figure 12.3).
Aeroponics:
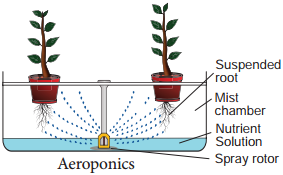
This technique was developed by Soifer Hillel and David Durger. It is a system where roots are suspended in air and nutrients are sprayed over the roots by a motor driven rotor (Figure 12.4).
Hydroponics and aeroponics are both methods of growing plants. The latter, aeroponics, is a method used to grow plants in the air – without the use of soil. Hydroponics is also a method that does not use soil, but instead, uses only a nutrient solution in a water solvent.
An advanced form of hydroponics, aeroponics is the process of growing plants with only water and nutrients. This innovative method results in faster growth, healthier plants, and bigger yields, all while using fewer resources. Plants grow in a soilless medium called rockwool.
In hydroponics we provide a solution in which plants can feed the amount they need when they want to. Because of this, we have more control over the speed and growth of our plants. Although this is a relatively new method of growing cannabis, it is cheaper than aeroponics.
Hydroponics offers the advantage of no energy wasted searching for nutrients. Aeroponic systems are a specialized version of hydroponics where the roots of the plant extend only in air and the roots are directly sprayed with a nutrient water mix (the recipe).
There are two main types of aeroponic systems: high pressure aeroponics and low pressure aeroponics. The main difference being the droplet size of the mist used in each case. Low-pressure aeroponics uses low-pressure, high-flow pumps, whereas high-pressure aeroponics uses high-pressure, low-flow pumps.
There are six main types of hydroponic systems to consider for your garden: wicking, deep water culture (DWC), nutrient film technique (NFT), ebb and flow, aeroponics, and drip systems.
The major disadvantage of aeroponics is the cost. Aeroponic systems are more expensive than most hydroponic systems and are completely dependent on a power source to run the air and nutrient pumps and the timer. Even a short interruption in power can result in the roots drying out and killing your plants.
One of the best advantages of Aeroponics is that plants grow quickly in such systems. They thrive and are able to produce great harvests. The plants grown are also much stronger and healthier due to this oxygen richness too.
In Aeroponic farming, the plantations can be vertical in the structure which helps the farmers to save a lot of space which in turn produces more food. There would be a great reduction in the disease and the infestation of pests. The Aeroponic farming can also be automated which reduces the cost of labour.
Fruits and Vegetables can also be grown comfortably in Aeroponics systems. Lot of vegetables and fruits can be grown like Beets, Broccoli, Cabbage, Carrots, Cauliflower, Corn, Cucumber, Eggplant, Grapes, Melons, Onions, Peas, Peppers, Potatoes, Radish, Raspberry, Strawberry, Sweet Potato, Tomatoes, and Watermelon.
Both hydroponics and aquaponics have clear benefits over soil-based gardening: lessened, adverse environmental impacts, reduced consumption of resources, faster plant growth, and higher yields. Many believe that aquaponics is a better option over hydroponics when choosing a soilless growing system.
Is hydroponics really good for the environment? Yes, hydroponics is good not just for the environment, but for several other reasons such as higher yield, water conservation and the removal of pesticides and herbicides.
You will have to mix up advanced nutrients for aeroponics of the proper strength consisting all of the required nutrients in the proper proportions (depending upon what your plant needs for its growth). You should not miss the Growing Exotic Hydroponic Plants.
Plants grown through hydroponics and aeroponics have the advantage natural and unrestricted growth. This system has enabled the cultivation of numerous plants that were previously considered difficult or impossible to grow from cuttings, as it becomes possible to propagate them from a single stem cutting.
There are vast numbers of people who have heard of hydroponics, and the majority of those know that systems can be set up indoors. In hydroponics, we can now provide all the light we need to plants to help them grow, so in this case, no they don’t need sunlight.