NCERT Solutions for Class 8 English Honeydew Chapter 8 A Short Monsoon Diary are part of NCERT Solutions for Class 8 English. Here we have given NCERT Solutions for Class 8 English Honeydew Chapter 8 A Short Monsoon Diary.
| Board | CBSE |
| Textbook | NCERT |
| Class | Class 8 |
| Subject | English Honeydew |
| Chapter | Chapter 8 |
| Chapter Name | A Short Monsoon Diary |
| Category | NCERT Solutions |
NCERT Solutions for Class 8 English Honeydew Chapter 8 A Short Monsoon Diary
IMPORTANT PASSAGES FOR COMPREHENSION
Read the following extracts and answer the questions that follow choosing the correct options among the given ones :
I. Through the mist Bijju is calling to his sister. I can hear him running about on the hillside but I cannot see him. (Page 113)
Multiple Choice Questions
1. The scene being described here is
(a) the first day of monsoon
(b) the last day of monsoon
(c) an ordinary day on the hills
(d) a winter day.
2. The author cannot see Bijju because
(a) he is not in the line of his eyes
(b) it is misty
(c) it is night
(d) he is blind.
3. The passage is written by
(a) Satyajit Ray
(b) Rabindranath Tagore
(c) Nathaniel Hawthorne Answers
(d) Ruskin Bond.
Answers:
1. (a) the first day of monsoon
2. (b) it is misty
3. (d) Ruskin Bond
II. In the evening it attacked one of Bijju’s cows but fled at the approach of Bijju’s mother, who came screaming imprecations.
As for the leeches, I shall soon get used to a little bloodletting every day. (Page 114)
Questions
1. What does ‘it’ in the first line refer to ?
2. What causes the bloodletting ?
3. What did Bijju’s mother do ?
4. Find a word in the passage which means ‘curses’. Answers
Answers:
1. ‘It’ refers to the leopard.
2. The leeches cause the bloodletting.
3. Bijju’s mother chased the leopard away.
4. Imprecations.
III. It is a good sound to read by—the rain outside, the quiet within—and, although tin roofs are given to springing unaccountable leaks, there is a feeling of being untouched by, and yet in touch with, the rain.
Multiple Choice Questions
1. Here, the author is praising
(a) tin roofs
(b) rain
(c) springing
(d) leaks.
2. The ‘good sound’ is produced by
(a) rain only
(b) tin roofs only
(c) rain and tin roofs
(d) rain and leaks.
3. The word ‘springing’ here means
(a) jumping
(b) creating
(c) a season
(d) finishing.
Answers:
1. (a) tin roofs
2. (c) rain and tin roofs
3. (b) creating
IV. The blackest cloud I’ve ever seen squatted over Mussoorie, and then it hailed marbles for half an hour. Nothing like a hailstorm to clear the sky. Even as I write, I see a rainbow forming. (Page 117)
Questions
1. What season is being described ?
2. What do the ‘marbles’ refer to ?
3. What does the author mean by ‘clear sky’ ?
Answers:
1. Rainy season is being described here.
2. The marbles refer to ‘hails’.
3. A clear sky means ‘a sky free of clouds’.
TEXTUAL EXERCISES
COMPREHENSION CHECK (Page 115)
1. Why is the author not able to see Bijju ?
2. What are the two ways in which the hills appear to change when the mist comes up ?
Answers
1. The author is not able to see Bijju because of the mist. The mist has blocked the vision.
2. The mist covers the hills and all the atmosphere. So they cannot be seen. The second change is that the birds stop singing. This makes the hills suddenly silent.
COMPREHENSION CHECK (Page 117)
1. When does the monsoon season begin and when does it end ? How do you prepare to face the monsoon ?
2. Which hill-station does the author describe in this diary entry ?
3. For how many days does it rain without stopping ? What does the author do on these days ?
4. Where do the snakes and rodents take shelter ? Why ?
5. What did the author receive in the mail ?
Answers
1. The monsoon generally begins in the end of June. It generally ends in the end of August or in the beginning of September. We buy raincoats or umbrellas to face the monsoon.
2. Mussoorie.
3. It has rained for eight or nine days without stopping. During these the author has been pacing the room or looking out of the window.
4. The snakes and rodents take shelter in roofs, attics and godowns. They do so because their holes are flooded with water. Out of the holes, these are the convenient places for them.
5. The author received a cheque in the mail.
WORKING WITH THE TEXT (Page 118)
Question. 1.
Look carefully at the diary entries for June 24-25, August 2 and March 23. Now write down the changes that happen as the rains progress from June to March.
Answer:
Monsoon arrives on June 24. The first real monsoon shower comes on June 25. The whole nature welcomes it. The author feels the joy himself and in everything around him.
By August 2, the people become weary of the monsoon. It is so because the movement in the open has become difficult. There is no sunshine for more than a week.
March 23 heralds the end of winter. After a hailstorm, there is a rainbow in the sky. It shows the beauty of nature.
Question. 2.
Why did the grandmother ask the children not to kill the Chuchundar ?
Answer:
Grandmother believed that Chuchundars are lucky. She thought that with them came money. So she asked the children not to kill it.
Question. 3.
What signs do we find in Nature which show that the monsoons are about to end ?
Answer:
At the end of the monsoon, the lush monsoon growth reaches its peak. The seeds of the cobra lily turn red. These things show that the monsoons are about to end.
Question. 4.
Complete the following sentences.
(i) Bijju is not seen but his voice is heard because___.
(ii) The writer describes the hill station and valley as___.
(iii) The leopard was successful in but had to flee when___.
(iv) The minivets are easily noticed because___.
(v) It looks like a fashion display on the slopes when___.
(vi) During the monsoon season, snakes and rodents are found in roofs and attics because___.
Answer:
(i) the mist obstructs the author’s vision.
(ii) ‘A paradise that might have been’.
(iii) killing a dog but had to flee when Bijju’s mother arrived crying curses.
(iv) of their bright colours.
(v) they are covered by a variety of flowers.
(vi) their holes are flooded with water and these things provide them convenient shelters.
Question. 5.
‘Although tin roofs are given to springing unaccountable leaks, there is a feeling of being untouched by, and yet in touch with, the rain.’
(i) Why has the writer used the word, ‘springing’ ?
(ii) How is the writer untouched by the rain ?
(iii) How is the writer in touch with the rain at the same time ?
Answer:
(i) The word ‘springing’ gives the idea of suddenness.
(ii) The author is untouched by the rain because he is in a room. The room pro-tects him from rain.
(iii) The writer hears the sound of rainfall on his tin-roof. So he is in touch with the rain.
Question. 6.
Mention a few things that can happen when there is endless rain for days together.
Answer:
When there is endless rain for days together, the life becomes difficult. To go out becomes difficult. One gets bored living in the room all the time. Everything becomes damp and soggy. The washed clothes do not get dried. Constant rainfall is an invitation for many insects, rodents and snakes to enter the house.
It can also cause diseases. The sun is the source of life. If sunlight doesn’t reach people for a long time, they fall ill.
Question. 7.
What is the significance of cobra lily in relation to the monsoon season, its beginning and end ?
Answer:
The first cobra lily is seen with the arrival of the monsoon. When its seeds begin to turn red, it is an indication that the rains are over.
WORKING WITH LANGUAGE (Page 118)
Question 1.
Here are some words that are associated with the monsoon. Add as many words as you can to this list. Can you find words for these in your languages ?
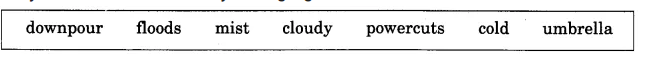
Answers
rainwater, fog, overcast, damp, soggy, raincoat, darkness, leaks.
Hindi words for those given in the question are :
![]()
Question 2.
Look at the sentences below.
(i) Bijju wandered into the garden in the evening.
(ii) The trees were ringing with birdsong.
Notice the highlighted verbs.
The verb wandered tells us what Bijju did that evening. But the verb was ringing tells us what was happening continually at same time in the past (the birds were chirping in the trees).
Now look at the sentences below. They tell us about something that happened in the past. They also tell us about other things that happened continually at the same time in the past.
Put the verbs in the brackets into their proper forms. The first one is done for you.
(i) We (get out) of the school bus. The bell (ring) and everyone (rush) to class.
We got out of the school bus. The bell was ringing and everyone was rushing to class.
(ii) The traffic (stop). Some people (sit) on the road and they (shout) slogans.
(iii) I (wear) my raincoat. It (rain) and people (get) wet.
(iv) She (see) a film. She (narrate) it to her friends who (listen) carefully.
(v) We (go) to the exhibition. Some people (buy) clothes while others (play) games.
(vi) The class (is) quiet. Some children (read) books and the rest (draw).
Answers:
(ii) The traffic stopped. Some people were sitting on the road and they were shouting slogans.
(iii) I was wearing my raincoat. It was raining and people were getting wet.
(iv) She had seen a film. She was narrating it to her friends who were listening carefully.
(v) We went to the exhibition. Some people were buying clothes while others were playing games.
(vi) The class was quiet. Some children were reading books and the rest were drawing.
Question 3.
Here are some words from the lesson which describe different kinds of sounds.

(i) Match these words with their correct meanings.
(a) to fall in small drops
(b) to make a sound by hitting a surface repeatedly
(c) to move quickly through the air, making a soft sound id) harsh sound made by birds
(e) ringing sound (of a bell or breaking glass, etc.)
(ii) Now fill in the blanks using the correct form of the words given above.
(a) Ramesh__on his desk in impatience.
(b) Rain water___from the umbrella all over the carpet.
(c) The pony___its tail.
(d) The___of breaking glass woke me up.
(e) The___of the raven disturbed the child’s sleep.
Answers:
(i) (a) —> drip
(b) —> drum
(c) —> swish
(d) —> caw
(e) —> tinkle
(ii) (a) Ramesh drummed on his desk in impatience.
(b) Rain water dripped from the umbrella all over the carpet.
(c) The pony swished its tail.
(d) The tinkle of the breaking glass woke me up.
(e) The caw of the raven disturbed the child’s sleep.
Question 4.
And sure enough, I received a cheque in the mail.
Complete each sentence below by using appropriate phrase from the ones given below.

1. I saw thick black clouds in the sky. And ___ ___ it soon started raining heavily.
2. The blue umbrella was ___ ___ for the brother and sister.
3. The butterflies are ___ ___ to get noticed
4. The lady was ___ ___ to chase the leopard
5. The boy was ___ ___ to call out to his sister.
6. The man was ___ ___ to offer help.
7. The victim’s injury was ___ ___ for him to get admitted in hospital
8. That person was ___ ___ to repeat the same mistake again.
9. He told me he was sorry and he would compensate for the loss. I said,___ ___
Answers:
(i) sure enough
(ii) big enough
(iii) colourful enough
(iv) brave enough
(v) anxious enough
(vi) kind enough
(vii) serious enough
(viii) foolish enough
(ix) “Fair enough”
SPEAKING (Page 120)
Question 1.
Do you believe in superstitions ? Why, or why not ? Working with your partner, write down three superstitious beliefs that you are familiar with.
Answer:
No. I don’t believe in superstitions. However there is one snag in this statement. One man’s belief may be superstitious to the other and vice versa.
Three common superstitions are :
(i) No. 13 is ominous
(ii) Stop if a black cat crosses your path.
(iii) Do not sleep with your feet pointing towards south.
Question 2.
How many different kinds of birds do you come across in the lesson ? How many varieties do you see in your neighbourhood ? Are there any birds that you used to see earlier in your neighbourhood but not now ? In groups discuss why you think this is happening.
Answer:
We come across five kinds of birds in this lesson. These are: minivets, drongos, crow, whistling thrush and ‘shrew’. We do not see many varieties in our neighbourhood. There are two birds which used to be seen in our neighbourhood but have now disap-peared. These are peacocks and vultures. Poaching, felling of trees and pollution of the environment are three important reasons for its happening.
WRITING (Page 121)
Question 1.
The monsoons are a time of great fun and even a few adventures: playing in the rain and getting wet, wading through knee-deep water on your way to school, water flooding the house or the classroom, powercuts and so on.
Write a paragraph describing an incident that occurred during the rains which you can never forget.
Or
Write a poem of your own about the season of spring when trees are in full bloom.
Answers:
A rainy day in summer is a great blessing. Last Friday, I had not yet left for school, when it began to rain. It rained heavily. Going to school was out of question. So I placed my books on the table and stood near the window. Water had collected everywhere. People were wading through water with raincoats on. Some had umbrellas in their hands. But these gave them little shelter. Children absented themselves from schools.
They played in rain-water and cared little for their mothers who got angry. The roads became slippery. Some persons who slipped on the road, spoiled their clothes. The children had a hearty laugh.
Or
Spring is the season of joy
When Nature becomes a toy.
The trees are in full bloom
Some flowers are in my room.
In spring comes Holi
Let’s play with colour and roli.
There is summer in the air
How lovely the birds and hare !
Spring is the season of joy
When Nature becomes a toy
The trees are in full bloom
I have roses in my room.
We hope the NCERT Solutions for Class 8 English Honeydew Chapter 8 A Short Monsoon Diary help you. If you have any query regarding NCERT Solutions for Class 8 English Honeydew Chapter 8 A Short Monsoon Diary, drop a comment below and we will get back to you at the earliest.