Find free online Chemistry Topics covering a broad range of concepts from research institutes around the world.
Avogadro’s number | Definition & Units
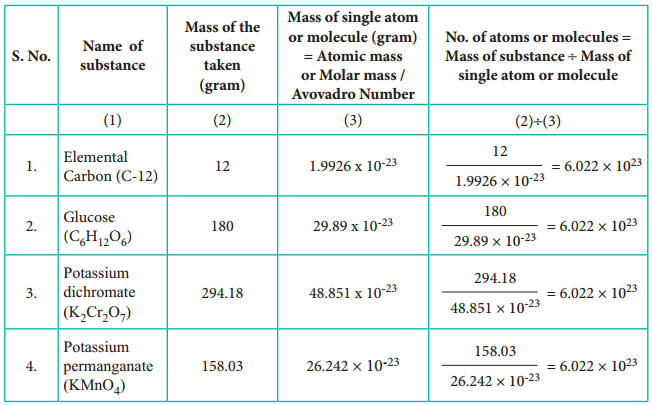
The total number of entities present in one mole of any substance is equal to 6.022 × 1023. This number is called Avogadro number which is named after the Italian physicist Amedeo Avogadro who proposed that equal volume of all gases under the same conditions of temperature and pressure contain equal number of molecules. Avogadro number does not have any unit.
In a chemical reaction, atoms or molecules react in a specific ratio. Let us consider the following examples
Reaction 1:
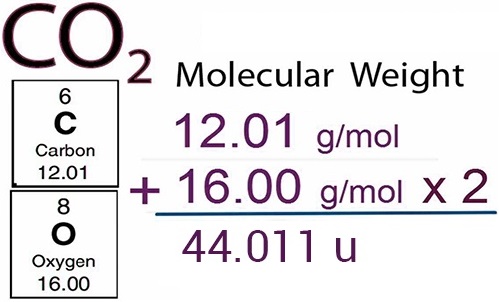
C + O2 → CO2
Reaction 2:
CH4 + 2O2 → CO2 + 2 H2O
In the first reaction, one carbon atom reacts with one oxygen molecule to give one carbon dioxide molecule. In the second reaction, one molecule of methane burns with two molecules of oxygen to give one molecule of carbon dioxide and two molecules of water.
It is clear that the ratio of reactants is based on the number of molecules. Even though the ratio is based on the number of molecules it is practically difficult to count the number of molecules.
Because of this reason it is beneficial to use ‘mole’ concept rather than the actual number of molecules to quantify the reactants and the products. We can explain the first reaction as one mole of carbon reacts with one mole of oxygen to give one mole of carbon dioxide and the second reaction as one mole of methane burns with two moles of oxygen to give one mole of carbon dioxide and two moles of water. When only atoms are involved, scientists also use the term one gram atom instead of one mole.

Molar Mass:
Molar mass is defined as the mass of one mole of a substance. The molar mass of a compound is equal to the sum of the relative atomic masses of its constituents expressed in g mol-1.
Examples:
- Relative atomic mass of one hydrogen atom = 1.008 u
- Molar mass of hydrogen atom = 1.008 g mol-1
- Relative molecular mass of glucose = 180 u
- Molar mass of glucose = 180 g mol-1
Molar Volume:
The volume occupied by one mole of any substance in the gaseous state at a given temperature and pressure is called molar volume.
Conditions | Volume occupied by one mole of any gaseous substances (in litre) |
| 273 K and 1 bar pressure (STP) | 22.71 |
| 273 K and 1 atm pressure | 22.4 |
| 298 K and 1 atm pressure (Room temperature & Pressure (SATP) | 22.47 |